Viagra gibt es mittlerweile nicht nur als Original, sondern auch in Form von Generika. Diese enthalten denselben Wirkstoff Sildenafil. Patienten suchen deshalb nach viagra generika schweiz, um ein günstigeres Präparat zu finden. Unterschiede bestehen oft nur in Verpackung und Preis.
Umc.br
Support Care Cancer (2011) 19:1069–1077DOI 10.1007/s00520-011-1202-0
A systematic review with meta-analysis of the effectof low-level laser therapy (LLLT) in cancer therapy-inducedoral mucositis
Jan Magnus Bjordal & Rene-Jean Bensadoun &Jan Tunèr & Lucio Frigo & Kjersti Gjerde &Rodrigo AB Lopes-Martins
Received: 26 August 2010 / Accepted: 30 May 2011 / Published online: 10 June 2011# Springer-Verlag 2011
Results We found 11 randomised placebo-controlled trials
Purpose The purpose of this study is to review the effects
with a total of 415 patients; methodological quality was
of low-level laser therapy (LLLT) in the prevention and
acceptable at 4.10 (SD±0.74) on the 5-point Jadad scale. The
treatment of cancer therapy-induced oral mucositis (OM).
relative risk (RR) for developing OM was significantly (p=
Methods A systematic review and meta-analysis of randomised
0.02) reduced after LLLT compared with placebo LLLT (RR=
placebo-controlled trials of LLLT performed during chemother-
2.03 (95% CI, 1.11 to 3.69)). This preventive effect of LLLT
apy or radiation therapy in head and neck cancer patients.
improved to RR=2.72 (95% CI, 1.98 to 3.74) when onlytrials with adequate doses above 1 J were included. For
J. M. Bjordal (*)
treatment of OM ulcers, the number of days with OM grade 2
Centre for Evidence-Based Practice,
or worse was significantly reduced after LLLT to 4.38 (95%
Bergen University College-HiB,
CI, 3.35 to 5.40) days less than placebo LLLT. Oral mucositis
Moellendalsvn. 6,
severity was also reduced after LLLT with a standardised
5009 Bergen, Norwaye-mail:
[email protected]
mean difference of 1.33 (95% CI, 0.68 to 1.98) over placeboLLLT. All studies registered possible side-effects, but they
were not significantly different from placebo LLLT.
Service d'Oncologie Radiothérapique,
Conclusions There is consistent evidence from small high-
CHU de Poitiers, BP 577, 86021 Poitiers Cedex, France
quality studies that red and infrared LLLT can partly prevent
development of cancer therapy-induced OM. LLLT also
Grängesberg Dental Clinic,
significantly reduced pain, severity and duration of symp-
Grängesberg, Sweden
toms in patients with cancer therapy-induced OM.
L. FrigoUniversity of Cruzeiro do Sul,
Keywords Low-level laser therapy. Oral mucositis .
Sao Miguel Paulista, SP, Brazil
Cancer. Chemotherapy. Radiation therapy
K. GjerdeDepartment of Clinical Odontology, University of Bergen,Bergen, Norway
R. A. Lopes-Martins
Oral mucositis (OM) is a serious and acute side-effect for
Institute of Biomedical Sciences, University of São Paulo (USP),
patients undergoing cancer therapy. The frequency of its
São Paulo, Brazile-mail:
[email protected]
appearance varies with therapy and cancer type up to 100%in oral cancer patients receiving adjuvant chemotherapy or
R. A. Lopes-Martins
radiotherapy [, ].
Centro de Pesquisa Clínica em Biofotônica Aplicada às Ciências
OM has great impact on a patient´s well-being. It may
da Saúde, Universidade Nove de Julho,São Paulo, Brazil
necessitate modifications of treatment planning, suspension
Support Care Cancer (2011) 19:1069–1077
recommended in treatment guidelines [, Recommendednonpharmacological treatments are oral care with mouthrinse] and cryotherapy []. The latter may also be used for theprevention of OM occurrence. Pharmacological agents havelargely been used for palliative care and pain relief, and someare recommended by consensus in spite of lacking scientificevidence from randomised controlled trials. These pharma-cological agents include patient-controlled analgesia withmorphine in transplant patients with hematological malig-nancies and topical anaesthetics like lidocaine alone, or incombination with diphenhydramine []. More recently,pharmacological focus has been directed towards theprevention of ulceration and the drug palifermin, a humankeratinocyte growth factor that stimulates the prolifer-ation, migration, and differentiation of epithelial cells and isrecommended in patients undergoing stem cell transplanta-tions. In addition, amifostine is thought to inhibit harmfulreactive oxygen species release but the scientificevidence for this drug is sparse. More recently, pharmaco-logical focus has been directed towards the prevention of
Fig. 1 Quorum flow chart showing the stages of the reviewing
ulceration (palifermin and amifostine) but no single inter-
process and the number of studies filtered out at each stage
vention yet serves as a panacea for all phases of OM.
Low-level laser therapy (LLLT) is a local application of a
of therapy, need for opioid analgesics, and/or require enteral
monochromatic, narrow-band, coherent light source. LLLT is
or parenteral nutrition with an impact on patient's survival
recommended as a treatment option for OM in the MASCC
]. The additional cost of OM treatment for cancer
guidelines but with limitations due to heterogeneous laser
patients can be considerable [].
parameters and a lack of dosage consensus in the LLLT
Many interventions have been used in OM management,
literature. The action of LLLT is disputed, but a cytoprotective
but only a handful of interventions have sufficient scientific
effect before and during oxidative stress has been observed
support from positive results in controlled clinical trials to be
after pre-treatment with LLLT There is some support
Table 1 Trial characteristics
First column identifies trial by first author's last name and the publication year. Other columns represent: sample size (type of cancer therapy),laser wavelength in nm, laser output in mW, spot size in cm2 , dose in Joules, irradiation time per point, outcomes reported including mucositisseverity scales (WHO or OMI), pain and duration of OM in days and dichotomized overall results given by: (+) significantly in favour of LLLT or(−) non-significant between LLLT and placebo

Support Care Cancer (2011) 19:1069–1077
Table 2 Trial methodological quality scored with an "x" if the methodological criterion is fulfilled
Withdrawals handled
The first column identifies each trial by first author's last name and last two digits of the publication year. The total methodological score (Jadadscale max. score=5) for each trial is given in the last column
for this protective LLLT effect in humans too ], and a
as advised by Dickersin et al. [] for randomised
possible therapeutic window has also been identified for an
controlled clinical trials. Keywords were: low-level laser
anti-inflammatory effect of red and infrared LLLT ].
therapy, low-intensity laser therapy, low-energy laser ther-
Evidence-based treatment guidelines have been forwarded
apy, phototherapy, HeNe laser, IR laser, GaAlAs, GaAs,
from the World Association for Laser Therapy (WALT)
diode laser, NdYag, oral mucositis, and cancer. Hand
searching was also performed in national physiotherapy
doses of LLLT have been identified for osteoarthritis [],
and medical journals from Norway, Denmark, Sweden,
tendinopathies [], and neck pain With the increasing
Holland, England, Canada, and Australia. Additional
body of randomised controlled trials, there seems to be a
information was gathered from LLLT researchers in the
need for systematically reviewing the literature and quantify
possible LLLT effects of LLLT in both prevention andtreatment of cancer therapy-induced OM.
Inclusion criteria
The randomised controlled trials were subjected to the
Materials and methods
following six inclusion criteria:
Literature search
1. Diagnosis: oral mucositis in cancer patients induced
after chemotherapy or radiation therapy
A literature search was performed on Medline, Embase,
2. Treatment: LLLT with wavelengths of 632–1,064 nm,
Cinahl, PedRo, and the Cochrane Controlled Trial Register
treating the mucosa of the oral cavity
Fig. 2 Forest plot showing themeta-analysis results forprevention of OM occurrence byLLLT dose compared withplacebo. Trial results plotted onthe right-hand side indicateeffects in favour of LLLT, andthe combined effects are plottedas black diamonds for dosesabove 1 J, below 1 J and overallregardless of dose, respectively
Support Care Cancer (2011) 19:1069–1077
Fig. 3 Forest plot showing themeta-analysis results forprevention of OM occurrence byLLLT wavelengths comparedwith placebo. Trial resultsplotted on the right-hand sideindicate effects in favour ofLLLT, and the combined effectsare plotted as black diamondsfor red wavelengths(630–670 nm), infraredwavelengths (780–830 nm), andoverall regardless ofwavelength, respectively(published online only)
3. Design: randomised parallel group design or crossover
5. Subgroup analyses were planned for (1) doses of <1 J
and >1 J (minimum dose according to WALT guidelines
4. Blinding: outcome assessors should be blinded
for other inflammatory conditions), (2) red and infrared
5 Control group: receiving identical placebo laser
wavelengths with their anticipated optimal dose ranges
6. Specific endpoints for prevention of oral mucositis
(1–4 J for red wavelengths and 3–8 J for infrared
above a certain grade, oral mucositis severity, duration
in days, and pain intensity
A statistical meta-analysis software package developed
by Cochrane Collaboration (Revman 5.0.22) was usedfor the statistical calculations. If heterogeneity was
1. The relative risk (RR) over placebo for preventing
present in heterogeneity tests, a random effects model
occurrence of oral mucositis above a certain grade (0–
was used for calculations. If heterogeneity was absent, a
fixed effects model was used for calculation of the overall
2. The effect of LLLT on the severity of oral mucositis
measured by the Oral Mucositis Index (OMI) or WHOscales were calculated as the SMD versus placebo.
Analysis of bias, including methodological quality, funding
3. The effect of LLLTon the duration of days oral mucositis
source, and patient selection
was calculated as the weighted mean difference versusplacebo
Positive bias direction, caused by flaws in trial
4. The effect of LLLT on pain intensity was calculated as
methodology, funding source
the standardised mean difference (SMD) versus placeboand labelled after Cohen [as "poor" (0.2–0.5),
Trials were subjected to methodological assessments by the
"good" (0.5–0.8), or "very good" (>0.8)
5-point Jadad checklist [For-profit funding sources
Fig. 4 Forest plot showing the meta-analysis results for duration of
LLLT, and the combined effect including variance is plotted as a black
OM after LLLT compared with placebo as a weighted mean difference.
diamond at the bottom of the forest plot
Trial results plotted on the right-hand side indicate effects in favour of
Support Care Cancer (2011) 19:1069–1077
Fig. 5 Forest plot showing the meta-analysis results for LLLT effect
results plotted on the right-hand side indicate effects in favour of
on OM severity compared with placebo as a standardised mean
LLLT, and the combined effect including variance is plotted as a black
difference (combines results from different OM severity scales). Trial
have been shown to affect trial conclusions in a positive
severity. The characteristics of the included trials and laser
direction [which made us include an analysis of
parameters are listed in Table .
funding sources. Methodological assessments were madeindependently according to the Jadad 5-point scale by two
Methodological quality
of the authors (JMB and RABLM).
The assessors gave similar methodological gradings for allthe included studies, and a consensus meeting was not
needed. Methodological quality was high for the includedstudies with a mean score of 4.10 (SD±0.74). The
Literature search and exclusion procedure
individual method scores are given in Table .
The literature search revealed 149 papers for oral mucositis
Funding sources analysis
and laser therapy. Thirty-three were regarded as potentiallyrelevant papers. Of these, nine studies were reviews and six
Laser manufacturers were acknowledged for support in two
studies were case studies while another three were animal
trial reports [One trial report explicitly stated that
studies. Three controlled studies were excluded for lack of
no conflicts of interest existed ] while another trial stated
randomization while one study lacked a placebo-control
that funding came from an independent non-profit source
group ]. The exclusion/inclusion procedure is described
]. Six trials did not explicitly mention conflicts of
according to the ] Quorum standard in Fig.
interest in their trial reports. But none of the affiliations and
The final sample consisted of 11 randomised placebo-
addresses in these reports indicated industry involvement.
controlled trials published from 1997 until 2009 with a total
Double checking the "Instructions to Authors" in the
of 415 patients []. The OMI was used in seven trials
journals in which these trial reports appeared, revealed that
and the WHO was used in one trial as measures of OM
the journals demanded declarations from the authors about
Fig. 6 Forest plot showing the subgroup meta-analysis results for
right-hand side indicate effects in favour of LLLT, and the combined
infrared LLLT doses of ≤2 J or >2 J of the effect on OM severity
effect including variance is plotted as a black diamond (published
compared with placebo as a standardised mean difference (combines
results from different OM severity scales). Trial results plotted on the
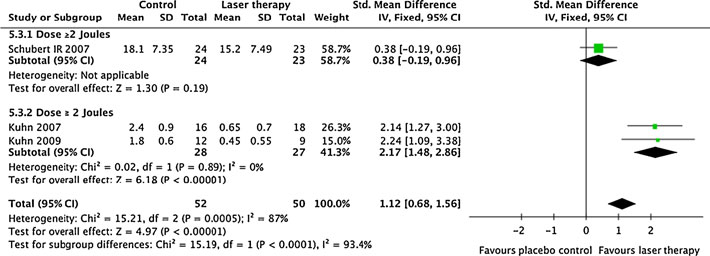
Support Care Cancer (2011) 19:1069–1077
Fig. 7 Forest plot showing the meta-analysis results for LLLT effect
the right-hand side indicate effects in favour of LLLT, and the
on pain compared with placebo as a standardised mean difference
combined effect including variance is plotted as a black diamond
(combines results from different pain scales). Trial results plotted on
(published online only)
possible conflicts of interest before publication. For this
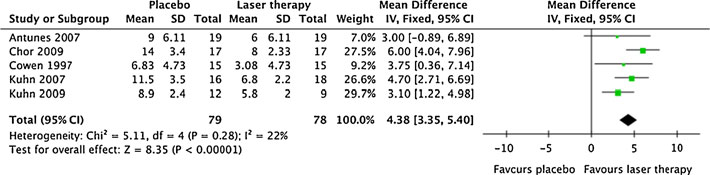
Effect on duration of oral mucositis
reason, the lack of mention has been accepted by theauthors as a lack of conflicts of interest, rather than
Five studies presented data for this outcome, and LLLT
undeclared conflicts of interest. In total, bias from for-
reduced significantly the number of days with oral
profit funding sources occurred in just two of 11 papers
mucositis grade 2 or worse with 4.38 (95% CI, 3.35 to
which the authors consider has negligible influence on the
5.40) days. The results for each individual study and the
review conclusion.
combined results are summarised in Fig.
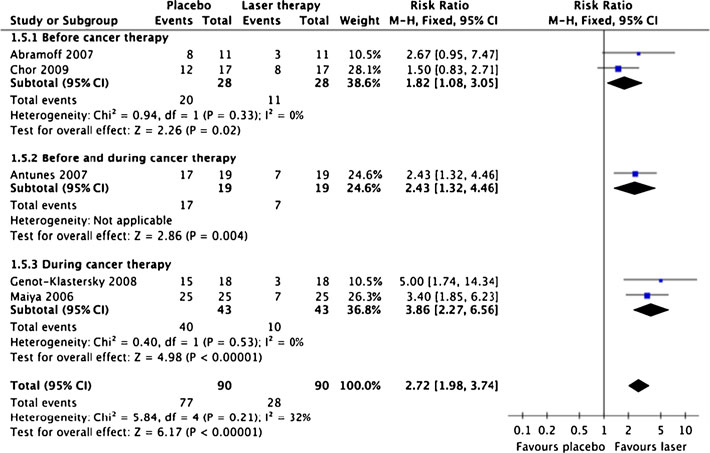
Relative risk for occurrence of cancer therapy-induced OM
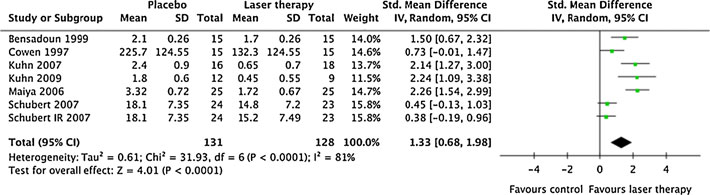
Effect on mucositis severity
Six trials presented seven different comparisons of contin-
Six studies started LLLT before OM ulcers occurred and
uous data for mucositis severity. As the trials used different
presented categorical data for the risk of developing OM
mucositis index scales, the combined results were calculat-
above a certain grade (OM grades 0, 1, 2) during cancer
ed as the SMD. The combined SMD effect size was 1.33
therapy. There was a significant preventive effect of LLLT
(95% CI, 0.68 to 1.98) and heterogeneity was present (p<
with a relative risk at 2.03 (95% CI, 1.11 to 3.69) less for
0.0001 and I2=81%). The results for each trial and the
cancer therapy-induced OM to occur. The analysis revealed
combined effect size are presented in Fig.
significant heterogeneity (I2=54%, p=0.03) between trials,and the results are summarised in Fig.
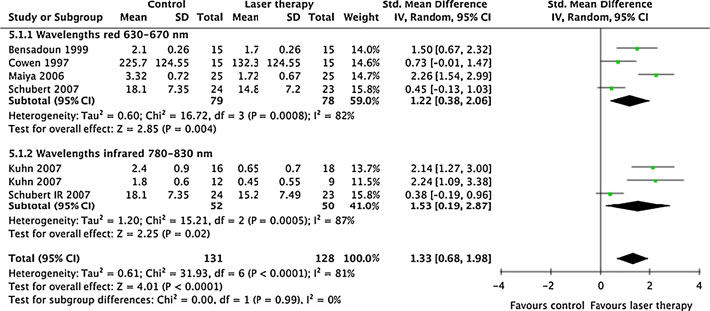
Dose analyses of anticipated optimal dose ranges
Analysis of irradiation parameters revealed that one
study ] had given a lower dose (0.18 J) than theminimum recommended WALT dose of 1 J. After sub-
A subgroup analysis of anticipated optimal dose ranges for red
grouping trials with doses above 1 J, heterogeneity
and infrared wavelengths on OM severity, revealed that
disappeared (I2=16%, p=0.31). The relative risk for
infrared wavelengths (6 J in both trials) gave an SMD at 2.17
preventing OM to occur increased to 2.72 (95% CI, 1.98
(95% CI, 1.48 to 2.86) without signs of heterogeneity between
to 3.74). The results for each study subgrouped by their
trials (I2=0% and p=0.89). A dose of 2 J with an infrared
timing of LLLT subgroups and the total RR are presented
wavelength was ineffective SMD 0.38 (95% CI, −0.19 to
0.96) in reducing mucositis severity. The dose analyses arepresented in Fig.
Subgroup analysis of LLLT wavelength effectson the relative risk for occurrence of OM after LLLT
Effect on pain relief
The subgroup analysis revealed no heterogeneity between
Four trials reported continuous data on pain intensity from
trials with anticipated optimal doses for the red (630–
different scales. The combined analysis revealed a signif-
670 nm) and the infrared (780–830 nm) subgroups,
icant effect of LLLT with an SMD at 1.22 (95% CI, 0.19 to
respectively (p>0.21 and I2<32%), and there were no
2.25) but also significant heterogeneity caused by one trial
significant wavelength differences in relative risks between
]. Removal of this study restored homogeneity (I2=0%
red and infrared at 2.72 (95% CI, 1.98 to 3.74) and infrared
and p=0.58), but reduced the effect size to 0.61 (95% CI,
at 3.48 (95% CI, 1.79 to 6.75).
0.29 to 0.94) (see Fig. ).
Support Care Cancer (2011) 19:1069–1077
Table 3 Summarised recommended treatment parameters
Minimum no. days to start
LLLT before cancer therapy
Side effects of LLLT
previous studies found no significant differences betweenred wavelengths in this range ]. For infrared wave-
All the studies investigated possible side-effects, but none
lengths, 830 nm was used in all trials but one underdosed
found side-effects or adverse effects beyond those reported
trial []. Doses were also fairly consistent across trials
for placebo LLLT. Five trials reported explicitly that LLLT
ranging from 1 to 6 J except the underdosed trial finding
was well tolerated among patients.
no significant effect from a dose 0.18 J. Treatment timesper point varied considerably with the variation in laseroutputs, but at least 17 s of irradiation per point was need
to achieve beneficial results (median, 50 s). The number oftreatment session varied from 3 to 30 in this material, but
This systematic review has revealed moderate to strong
this heterogeneity must be seen in conjunction with the
evidence for the efficacy of LLLT in cancer therapy-
heterogeneity in durations of chemotherapy and radiation
induced OM. A possible limitation to our findings is the
therapy regimens. Our interpretation is that LLLT needs to
small sample size of the included trials. Our finding is
be performed at least every other day for the duration of
partly contradicting a Cochrane review [] which was
chemotherapy and radiation therapy regimens, or as long
recently updated [Our review deviates from their
as OM ulcers are present. The trials which aimed at the
conclusions because we have included more studies and
prevention of OM started LLLT at 7 days before chemo-
subgroup analyses by dose range and wavelengths. The
therapy/radiation therapy regimens. It should be a target
overall scientific quality of the trials was methodologically
for future trials to compare treatment start at different
acceptable, but the heterogeneous treatment procedures
timepoints before cancer therapy to avoid unnecessary
and dosing may cause confusion. In the MASCC guide-
lines, the evidence behind LLLT is characterized as
From the evidence, we propose a fairly simple procedure
promising, but it is added that conflicting evidence with
for diode lasers for prevention and treatment of cancer
large operator variability and expensive equipment (gas
therapy-induced OM. LLLT should be performed with a red
lasers) limits more widespread clinical use []. The lasers
or infrared diode laser with outputs of 10–100 mW in a
used in the studies reviewed are relatively inexpensive
stationary manner (not scanning). The parameters are
diode lasers (from $2,500) with low optical outputs (10–
summarised in Table .
100 mW), which have substituted the older more expen-
In manifested OM, lesions and inflammatory areas
sive gas lasers from the early LLLT trials [After
should be specifically targeted for irradiation. Our
reviewing the apparent discrepancies of the material, our
findings relate well to the emerging LLLT evidence of
subgroup analyses revealed plausible causes for the few
optimal doses in inflammatory conditions such as
conflicting results. A common misunderstanding in the
rheumatoid arthritis [] and acute postoperative pain
LLLT literature is caused by reporting clinical doses for
]. It is also interesting to note that the variety of
diode lasers with small spot sizes in Joules/cm2 rather than
different cancer therapies involved in the included trials
in Joules. If the spot size is very small, then the irradiation
did not seem to seriously interfere with the beneficial
time will be very short. This led to under-dosing in one of
effects of LLLT. How LLLT efficacy compares with the
the included trials, where they irradiated for 3 s per point
efficacy of pharmacological agents in OM, is outside the
WALT recommends that doses in clinical studies
scope for this review but this should certainly be a topic for
should be reported in Joules instead of Joules/cm2. LLLT
future research. In terms of side-effects, LLLT was well
wavelengths and doses were fairly homogeneous in the
tolerated and no serious incidents or withdrawals due to
other studies. Red wavelengths from 633 to 685 nm, and
treatment intolerance were reported.
Support Care Cancer (2011) 19:1069–1077
9. Lalla RV, Sonis ST, Peterson DE (2008) Management of oral
mucositis in patients who have cancer. Dent Clin North Am52:61–77, viii
We conclude that there is moderate to strong evidence in
10. Elting LS, Shih YC, Stiff PJ, Bensinger W, Cantor SB, Cooksley
favour of LLLT applied with doses of 1–6 J per point in the
C (2007) Economic impact of palifermin on the costs of
oropharyngeal area in cancer patients receiving chemotherapy
hospitalization for autologous hematopoietic stem-cell transplant:
or radiation therapy. There are limitations to the material in
analysis of phase 3 trial results. Biol Blood Marrow Transplant 13(7):806–813
terms of small sample size in the included trials. However, the
11. Hwang WY, Koh LP, Ng HJ, Tan PH, Chuah CT, Fook SC et al (2004)
material was consistently in favour of LLLT in both in the
A randomized trial of amifostine as a cytoprotectant for patients
prevention of OM occurrences and reductions of severity,
receiving myeloablative therapy for allogeneic hematopoietic stem
pain, and duration of OM ulcers.
cell transplantation. Bone Marrow Transpl 34:51–56
12. Lubart R, Eichler M, Lavi R, Friedman H, Shainberg A (2005)
Low-energy laser irradiation promotes cellular redox activity.
Acknowledgements Post-mortem to professor Anne Elisabeth
Photomed Laser Surg 23:3–9
Ljunggren for her encouragement and contribution to the early work
13. Rizzi CF, Mauriz JL, Freitas Correa DS, Moreira AJ, Zettler CG,
on this manuscript. Unfortunately, she was unable to see its
Filippin LI et al (2006) Effects of low-level laser therapy (LLLT) on
the nuclear factor (NF)-kappaB signaling pathway in traumatizedmuscle. Lasers Surg Med 38:704–713
Conflicts of interest None
14. Lopes-Martins RA, Marcos RL, Leonardo PS, Prianti AC Jr,
Muscara MN, Aimbire F et al (2006) Effect of low-level laser(Ga-Al-As 655 nm) on skeletal muscle fatigue induced byelectrical stimulation in rats. J Appl Physiol 101:283–288
15. Leal Junior EC, Lopes-Martins RA, Frigo L, De Marchi T, Rossi
RP, de Godoi V et al (2010) Effects of Low-level laser therapy(LLLT) in the development of exercise-induced skeletal muscle
1. Manas A, Palacios A, Contreras J, Sanchez-Magro I, Blanco P,
fatigue and changes in biochemical markers related to post-
Fernandez-Perez C (2009) Incidence of oral mucositis, its
exercise recovery. J Orthop Sports Phys Ther 40(8):524–532
treatment and pain management in patients receiving cancer
16. Bjordal JM, Johnson MI, Iversen V, Aimbire F, Lopes-Martins RA
treatment at Radiation Oncology Departments in Spanish hospitals
(2006) Photoradiation in acute pain: a systematic review of
(MUCODOL Study). Clin Transl Oncol 11:669–676
possible mechanisms of action and clinical effects in randomized
2. Elting LS, Keefe DM, Sonis ST, Garden AS, Spijkervet FK,
placebo-controlled trials. Photomed Laser Surg 24:158–168
Barasch A et al (2008) Patient-reported measurements of oral
17. Bjordal JM, Johnson MI, Lopes-Martins RA, Bogen B, Chow R,
mucositis in head and neck cancer patients treated with
Ljunggren AE (2007) Short-term efficacy of physical interventions in
radiotherapy with or without chemotherapy: demonstration of
osteoarthritic knee pain. A systematic review and meta-analysis of
increased frequency, severity, resistance to palliation, and
randomised placebo-controlled trials. BMC Musculoskelet Disord
impact on quality of life. Cancer 113:2704–2713
3. Elting LS, Cooksley C, Chambers M, Cantor SB, Manzullo E,
18. Bjordal JM, Lopes-Martins RA, Joensen J, Ljunggren AE,
Rubenstein EB (2003) The burdens of cancer therapy. Clinical and
Couppe C, Stergioulas A et al (2008) A systematic review with
economic outcomes of chemotherapy-induced mucositis. Cancer
procedural assessments and meta-analysis of low level laser
therapy in lateral elbow tendinopathy (tennis elbow). BMC
4. Nonzee NJ, Dandade NA, Patel U, Markossian T, Agulnik M,
Musculoskelet Disord 9:75
Argiris A et al (2008) Evaluating the supportive care costs of
19. Chow RT, Johnson MI, Lopes-Martins RA, Bjordal JM (2009)
severe radiochemotherapy-induced mucositis and pharyngitis:
Efficacy of low-level laser therapy in the management of neck pain: a
results from a Northwestern University Costs of Cancer Program
systematic review and meta-analysis of randomised placebo or
pilot study with head and neck and nonsmall cell lung cancer
active-treatment controlled trials. Lancet 374:1897–1908
patients who received care at a county hospital, a Veterans
20. Dickersin K, Scherer R, Lefebvre C (1994) Identifying relevant studies
Administration hospital, or a comprehensive cancer care center.
for systematic reviews. BMJ 309:1286–1291, 12 November 1994
Cancer 113:1446–1452
21. Cohen J (1977) Statistical power analysis for behavioural
5. Keefe DM, Schubert MM, Elting LS, Sonis ST, Epstein JB,
sciences. Academic, New York
Raber-Durlacher JE et al (2007) Updated clinical practice
22. Jadad AR, Moore RA, Carroll D, Jenkinson C, Reynolds DJM,
guidelines for the prevention and treatment of mucositis.
Gavaghan DJ et al (1996) Assessing the quality of reports of
Cancer 109:820–831
randomized clinical trials: is blinding necessary? Controlled Clin
6. Worthington HV, Clarkson JE, Eden OB (2007) Interventions for
preventing oral mucositis for patients with cancer receiving
23. Kjaergard LL, Als-Nielsen B (2002) Association between
treatment. Cochrane Database Syst Rev. CD000978
competing interests and authors' conclusions: epidemiological
7. Lanzos I, Herrera D, Santos S, O'Connor A, Pena C, Lanzos E et
study of randomised clinical trials published in the BMJ. BMJ
al (2010) Mucositis in irradiated cancer patients: effects of an
antiseptic mouthrinse. Med Oral Patol Oral Cir Bucal 15(5):e732–
24. Simoes A, Eduardo FP, Luiz AC, Campos L, Sa PH, Cristofaro M
et al (2009) Laser phototherapy as topical prophylaxis against
8. Lilleby K, Garcia P, Gooley T, McDonnnell P, Taber R, Holmberg
head and neck cancer radiotherapy-induced oral mucositis:
L et al (2006) A prospective, randomized study of cryotherapy
comparison between low and high/low power lasers. Lasers Surg
during administration of high-dose melphalan to decrease the
severity and duration of oral mucositis in patients with multiple
25. Shea B, Moher D, Graham I, Pham B, Tugwell P (2002) A comparison
myeloma undergoing autologous peripheral blood stem cell
of the quality of Cochrane reviews and systematic reviews published in
transplantation. Bone Marrow Transplant 37:1031–1035
paper-based journals. Eval Health Prof 25:116–129
Support Care Cancer (2011) 19:1069–1077
26. Antunes HS, Ferreira EM, de Matos VD, Pinheiro CT, Ferreira
33. Kuhn A, Porto FA, Miraglia P, Brunetto AL (2009) Low-level
CG (2008) The Impact of low power laser in the treatment of
infrared laser therapy in chemotherapy-induced oral mucositis: a
conditioning-induced oral mucositis: a report of 11 clinical cases
randomized placebo-controlled trial in children. J Pediatr Hematol
and their review. Med Oral Patol Oral Cir Bucal 13:E189–E192
27. Arun Maiya G, Sagar MS, Fernandes D (2006) Effect of low level
34. Schubert MM, Eduardo FP, Guthrie KA, Franquin JC, Bensadoun
helium-neon (He–Ne) laser therapy in the prevention & treatment
RJ, Migliorati CA et al (2007) A phase III randomized double-
of radiation induced mucositis in head and neck cancer patients.
blind placebo-controlled clinical trial to determine the efficacy of
Indian J Med Res 124:399–402
low level laser therapy for the prevention of oral mucositis in
28. Abramoff MM, Lopes NN, Lopes LA, Dib LL, Guilherme A,
patients undergoing hematopoietic cell transplantation. Support
Caran EM et al (2008) Low-level laser therapy in the prevention
Care Cancer 15:1145–1154
and treatment of chemotherapy-induced oral mucositis in young
35. Kuhn A, Vacaro G, Almeida D, Machado A, Braghini PB, Shilling
patients. Photomed Laser Surg 26:393–400
MA et al (2007) Low-level infrared laser therapy for chemo- or
29. Bensadoun RJ, Franquin JC, Ciais G, Darcourt V, Schubert MM, Viot
radiation-induced oral mucositis: a randomized placebo-controlled
M et al (1999) Low-energy He/Ne laser in the prevention of radiation-
study. J Oral Laser Applications 7:175–181
induced mucositis. A multicenter phase III randomized study in
36. Chor A, Torres SR, Maiolino A, Nucci M (2010) Low-power laser
patients with head and neck cancer. Support Care Cancer 7:244–252
to prevent oral mucositis in autologous hematopoietic stem cell
30. Cowen D, Tardieu C, Schubert M, Peterson D, Resbeut M,
transplantation. Eur J Haematol 4:78–79
Faucher C et al (1997) Low energy helium–neon laser in the
37. Clarkson JE, Worthington HV, Furness S, McCabe M, Khalid T,
prevention of oral mucositis in patients undergoing bone marrow
Meyer S (2010) Interventions for treating oral mucositis for
transplant: results of a double blind randomized trial. Int J Radiat
patients with cancer receiving treatment. Cochrane Database Syst
Oncol Biol Phys 38:697–703
Rev 8:CD001973.
31. Genot-Klastersky MT, Klastersky J, Awada F, Awada A, Crombez
38. Albertini R, Villaverde AB, Aimbire F, Salgado MA, Bjordal JM,
P, Martinez MD et al (2008) The use of low-energy laser (LEL) for
Alves LP (2007) Anti-inflammatory effects of low-level laser
the prevention of chemotherapy- and/or radiotherapy-induced oral
therapy (LLLT) with two different red wavelengths (660 nm and
mucositis in cancer patients: results from two prospective studies.
684 nm) in carrageenan-induced rat paw edema. J Photochem
Support Care Cancer 16:1381–1387
Photobiol B 89(1):50–55
32. Cruz LB, Ribeiro AS, Rech A, Rosa LG, Castro CGJ, Brunetto
39. Brosseau L, Robinson V, Wells G, Debie R, Gam A, Harman K
AL (2007) Influence of low-energy laser in the prevention of oral
et al. (2005) Low level laser therapy (classes I, II and III) for
mucositis in children with cancer receiving chemotherapy. Pediatr
treating rheumatoid arthritis. Cochrane Database Syst Rev
Blood Cancer 48:435–440
Source: http://www.umc.br/artigoscientificos/art-cient-0052.pdf
Brother™ PocketJet® 6 Mobile Thermal Printers Home Healthcare Case Study PocketJet® 6 mobile thermal printers provide a better alternative to inkjets for the Midwest Pal iative & Hospice CareCenter. "We selected Brother On the heels of this mandate, new Electronic Moving forward, Midwest CareCenter plans to PocketJet® mobile
LAB #: U������������ CLIENT #: ����� PERCENTILE per g creatinine 2.5th 16th 50th 84th 97.5th 3,4-Dihydroxyphenylacetic acid (DOPAC) 3-Methoxytyramine (3-MT) Norepinephrine, free Epinephrine, free 5-Hydroxyindolacetic acid (5-HIAA) Phenethylamine (PEA) <dl: less than detection limit









