Viagra gibt es mittlerweile nicht nur als Original, sondern auch in Form von Generika. Diese enthalten denselben Wirkstoff Sildenafil. Patienten suchen deshalb nach viagra generika schweiz, um ein günstigeres Präparat zu finden. Unterschiede bestehen oft nur in Verpackung und Preis.
Editorial.ucatolica.edu.co
Felipe Cortés-Salazar1, Josué Omar Suárez Ortíz1, Nancy Mónica Cendejas Trejo1,
Juan Manuel Mancilla-Díaz1, Verónica Elsa López-Alonso1, Rodrigo Erick Escartín-Pérez1*
1Neurobiology of Eating Laboratory, Universidad Nacional Autónoma de México, FES Iztacala. Tlalnepantla, México
Recibido, mayo 2/2014
Referencia: Cortés-Salazar, F., Suárez Ortíz, J.
Concepto de evaluación, mayo 12/2014
O., Cendejas Trejo, N. M., Mancilla-Díaz, J. M.,
Aceptado, junio 23/2014
López-Alonso, V. E. & Escartín-Pérez, R. E. (2014).
Effects of CB1 cannabinoid receptor activation in
the nucleus accumbens shell on feeding behavior.
Acta Colombiana de Psicología, 17(2): pp. 61-68.
Obesity and its related pathologies are well- known health hazards. Although obesity and overweight have multifactorial causes, overeating is common in both of these conditions. According to animal models, endocannabinoids and their receptors in the brain play a key role in the genesis and development of obesity. It has been proposed that the cannabinoid receptors CB1 (RCB1) expressed in the nucleus
accumbens shell (NAC) are involved in the increase of the hedonic properties of food. To test this hypothesis, this study aimed to assess the effects of activating the NACs RCB1 on standard food intake during the light phase of the light-dark cycle. The effects of activating the RCB1 with CP 55,940 and WIN 55-212-2 (0.125, 0.25 and 0.5 nmol) in the NACS on feeding behavior and the behavioral satiety sequence of rats were assessed. It was found that both agonists increased food intake and delayed expression of satiety during the light phase. These results suggest that cannabinoid agonists encourage food intake when motivation is low and palatability is normal.
Key words: Cannabinoids, food, nucleus accumbens shell, behavioral satiety sequence.
La obesidad y sus patologías relacionadas son riesgos de salud muy conocidos. Aunque la obesidad y el sobrepeso tienen causas multifactoriales, la sobreingesta de alimento es frecuente en estas condiciones. De acuerdo con modelos animales, los endocanabinoides y sus receptores en el cerebro juegan un papel clave en la génesis y desarrollo de la obesidad. Se ha propuesto que los receptores a canabinoides CB1 (RCB1) expresados en el núcleo
accumbens shell (NAcS) están involucrados en el incremento de las propiedades hedónicas del alimento. Para probar esta hipótesis, este estudio tuvo como objetivo evaluar los efectos de la activación de los RCB1 en el NAcS sobre la ingesta de alimento estándar durante la fase de luz del ciclo luz-oscuridad. Se evaluaron los efectos de la activación de los RCB1 con WIN 55-212-2 y CP 55,940 (0.125, 0.25, y 0.5 nmol) en el NAcS sobre la conducta alimentaria y la secuencia de saciedad conductual en ratas. Se encontró que ambos agonistas aumentaron la ingesta de alimento y demoraron la expresión de la saciedad durante la fase de luz. Lo anterior sugiere que los agonistas canabinoides estimulan el consumo de alimento cuando la motivación por el mismo es baja y la palatabilidad es normal. Palabras clave: Canabinoides, alimentación, núcleo
accumbens shell, secuencia de saciedad conductual.
* R. Erick Escartín-Pérez. Neurobiology of Eating Laboratory, UNAM FES Iztacala. 1 Av. de los Barrios, Los Reyes Iztacala, Tlalnepantla,
México, 54090, México. Tel: + (52 55) 56231333 ext 39717. FAX: + (52 55) 5390 Acknowledgements. Funding for this study was provided by UNAM DGAPA grant IN224811 and IN224214 and FESI PAPCA 15.
CORTÉS-SALAZAR, SUÁREZ ORTÍZ, CENDEJAS TREJO, MANCILLA-DÍAZ, LÓPEZ-ALONSO, ESCARTÍN-PÉREZ
A obesidade e suas patologias relacionadas são riscos de saúde muito conhecidos. Ainda que a obesidade e o sobrepeso possuam causas multifatoriais, a sobre ingestão de alimento é frequente nestas condições. De acordo com modelos animais, os endocanabinóides e seus receptores no cérebro jogam um papel chave na gênese e desenvolvimento da obesidade. Foi proposto que os receptores a canabinóides CB1 (RCB1) expressos no núcleo
accumbens shell (NAcS) estão envolvidos no aumento das propriedades hedônicas do alimento. Para testar esta hipótese, este estudo teve como objetivo avaliar os efeitos da ativação dos RCB1 nos NAcS sobre a ingestão de alimento padrão durante a fase de luz do ciclo luz-escuridão. Avaliaram-se os efeitos da ativação dos RCB1 com WIN 55-212-2 e CP 55,940 (0.125, 0.25, e 0.5 nmol) no NAcS sobre a conduta alimentar e a sequência de saciedade condutual em ratos. Encontrou-se que ambos agonistas aumentaram a ingestão de alimento e demoraram a expressão da saciedade durante a fase de luz. Isso sugere que os agonistas canabinóides estimulam o consumo de alimento quando a motivação pelo mesmo é baixa e a palatabilidade é normal. Palavras chave: Canabinóides, alimentação, núcleo
accumbens shell, sequência de saciedade conductual.
Obesity and its related pathologies are well-known health
the effects of activation of CB1R in the NAcS on feeding
threats. Despite obesity and overweight have multifactorial
behavior using the behavioral satiety sequence (BSS) analy-
causes, recurrent over-consumption of energy dense foods
sis. It is hypothesized that acute activation of CB1R in the
is frequently present in these conditions. Those diets are
NAcS will stimulate feeding when a standard chow food
commonly highly palatable and may activate the brain
is offered during the light phase of the photoperiod, when
reward circuit (Bassareo & Di Chiara, 1999; Mellis, et al.,
the basal food intake is naturally low and the hedonically
2007), promoting over-consumption when these foods are
positive sensory properties of food may be stimulated by
freely available (Perello, Chuang, Scott & Lutter, 2010) or
cannabinoid agonists.
with limited access schedules (Berner, Avena & Hoebel,
2008; Dimitriou, Rice & Corwin, 2000).
The nucleus
accumbens shell (NAcS) plays a key role
in processing the rewarding properties of stimuli, and sev-
eral neurochemical signaling pathways interact to regulate
A total of 38 male Wistar rats (UNAM, FESI México)
dopamine release in the NAcS and contribute to provide
weighting 220-240 g at the beginning sessions were used.
rewarding and/or hedonic properties to food (Gardner,
Rats were housed in clear plastic cages with an inverted
2005). The endocannabinoid system has been implicated
12x12 h light and dark cycle with standard food (LabDiet®
in reward-driven feeding (Matias, Cristino & Di Marzo,
formulab diet #5008) and water
ad libitum. All experimen-
2008), and interestingly cannabinoid receptor 1 (CB1R) KO
tal procedures complied with the Official Mexican Norm
mice are resistant to diet-induced obesity, indicating that
(NOM-062-ZOO-1999), entitled Technical Specifications
energy dense foods require endocannabinoid signaling to
for the Production, Care, and Use of Laboratory Animals.
generate obesity (Quarta, et al., 2010). The experimental
evidence indicates that the acute activation of CB1R in the
Instruments and materials
NAcS by administration of anandamide (Soria-Gómez, et
Drugs and treatments. CP 55 940 (non-selective CB1R
al., 2007) or 2-AG (Kirkham, Williams, Fezza & Di Marzo,
agonist) and WIN 55 212-2 (non-selective CB1R agonist)
2002) consistently stimulates chow intake.
(Tocris Cookson Inc., Ellisville, MO, USA). Both drugs
Previous results in our laboratory showed that activation
were dissolved in DMSO, Tween-80 and 0.9 % saline
of CB1R in the paraventricular nucleus of the hypothalamus
solution (4:2:94 v/v/v). Different doses of drugs were
prevented the natural satiety sequence by modulating homeo-
administered intra-NAcS according to the experimental
static mechanisms (Escartín-Pérez, et al., 2009); nevertheless
design in a volume of 0.5 μl at 0.2 μl/min. Fresh solutions
activation of cannabinoid receptors in the NAcS might also
of drugs were prepared before the injections.
stimulate feeding by a different mechanism, for instance
Behavioral test (food intake and behavioral satiety
stimulating the hedonically positive sensory properties of
sequence). Consumption of standard chow (g) was as-
food. Accordingly, the present study was aimed to evaluate
sessed in two independent periods of 60 min each (hour 1,
food intake stimulated by cannabinoids
hour 2), 10 and 70 min post injection. The analysis of the
and they had a surgical recovery period of five days with
BSS was conducted in both 60 min observation periods,
food and water ad libitum.
which were divided in 12 segments (5 min bins). The
Design and recordings. After the surgical recovery
duration (s) of each behavior was measured from the
period, animals were randomly assigned to independent
videotape recording to the following mutually exclusive
groups that received vehicle or different intra-NAcS treat-
categories: Intake (standard food), resting (inactivity with
ments of WIN 55 212-2 (0.125, 0.25, 0.5 nmol) or CP 55
or without closed eyes, head of the rat was on the cage
940 (0.125, 0.25, 0.5 nmol) and behavioral recordings were
floor), activity (any other behavior different to feeding,
conducted. Immediately after central injections, animals
resting, and grooming including locomotion, smelling,
were placed into their home cages with pre-weighted stan-
sniffing, etc), and grooming (self-cleaning of any part
dard chow; behavior was video-recorded and subsequently
of the body). Durations of each behavioral category are
measured to obtain the BSS during two consecutive hours
presented in terms of duration (±SEM) over 12 periods
(two recordings of 60 min each, hour 1 and hour 2). At the
(5-min bins) per hour, and served as the measure of the
end of the observations, food containers were removed
progression of BSS (Escartín-Pérez, et al., 2009).
and ingestion of food was calculated (g). Animals were
Histology. After the behavioral tests, rats were given
individually housed with food and water ad libitum. All
a lethal dose of sodium pentobarbital and were beheaded,
behavioral observations were conducted during the light
brains were removed and fixed in 10% formalin for two
phase of the light/dark cycle.
days. Brains were sectioned at 100 μm in the coronal
Statistical analysis. Analyses of data from food intake
plane, and then positions of the cannulas and injection
measurements were performed by two-way ANOVAs (dose
sites were verified. Brains in which drugs were not ad-
x hour) to calculate significance of the differences by intra-
ministered in the NAcS were excluded from the present
NAcS injections. When appropriate, the Bonferroni's test
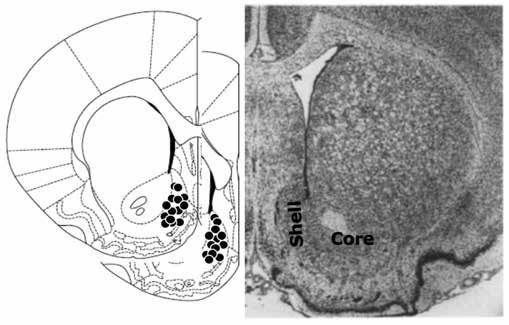
report (Figure 1).
was performed. The criterion for statistical significance
was p < 0.05. Data were analyzed using GraphPad Prism Version 5.0.
Data from standard food consumption measurements
(g) and the BSS (s) are expressed as means (± SEM) and are organized in two independent sections: 1) Food intake, and 2) Behavioral satiety sequence (BSS) analyses. Effects of cannabinoid receptors activation in the NAcS on food intake
Firstly, it was investigated if intra-NAcS activation of
Figure 1. Schematic representation of injection sites (black circles,
cannabinoid receptors with two different agonists produced
left) in the NAcS.
significant changes in chow consumption in a situation
where the basal intake is low (light phase of photoperiod).
Consistent with reports that found a stimulatory effect of
Surgery. All animals were anaesthetized with ketamine
activation of CB1R in the nucleus accumbens, intra-NAcS
and xilasine (112.5 mg/kg and 22.5 mg/kg I.P.) and were
injections of WIN 55 212-2 (all doses) significantly increased
stereotaxically implanted (unilaterally) with guide cannulas
standard chow intake (interaction dose x hour F
for injection in the nucleus accumbens shell (coordinates
factor dose F = 7.511; factor hour F = 25.81;p< 0.05)
AP +1.52 mm and ML -0.7 mm relative to bregma, DV
during the first hour of observation, and the effect persisted
-6.0 mm relative to dura mater) according to Paxinos and
during the second hour of the behavioral record when a
Watson's Atlas (1998). After surgery, animals were
dose of 0.25 nmol was administered (Figure 2).
admin- istered with benzathine penicillin (300,000 UI/kg, I.M.)
CORTÉS-SALAZAR, SUÁREZ ORTÍZ, CENDEJAS TREJO, MANCILLA-DÍAZ, LÓPEZ-ALONSO, ESCARTÍN-PÉREZ
Effects of cannabinoid receptors activation in the NAcS on the behavioral satiety sequence
In order to assess the behavioral profile associated to
the stimulatory effect of intra-NAcS cannabinoid receptor activation on chow intake, the BSS in the same periods of time (hour 1 and hour 2) was analyzed. It was found that control animals began the test eating during the first periods, but subsequently the time spent eating decreased while the time of resting increased, showing the transition from eating to resting after the third period of the first hour of observation; activity and grooming remained low dur- ing the complete test (hour 1 and hour 2). As expected, in the second hour, control animals spent time resting during almost the complete 60-min record, reflecting satiety (Fig.
4. A, B). Animals treated with 0.125 and 0.25 nmol of WIN
Figure 2. Effects of intra-NAcS injections of different doses of WIN
55 212-2 showed a delayed transition from eating to rest-
55 212-2 (WIN) on standard chow intake during two behavioral re-
ing in the first hour of observation (6th and 7th periods,
cords (hour 1 and hour 2). Data expressed in terms of means ± SEM.
respectively), resting less time and with a higher level of
*p<0.05, ***p<0.001 vs vehicle group (Vh); n= 4-6 per group.
activity (specially the 0.125 nmol dose) than control rats.
During the second hour of observation, animals treated with
Independent groups of rats were administered with the
the lower dose of WIN 55 212-2 (0.125 nmol) remained
non-selective cannabinoid receptor agonist, CP 55 940,
comparable to the control group; however, the 0.25 nmol
at different doses. It was found that the activation of can-
dose induced new meals and a higher level of activity,
nabinoid receptors in the NAcS (all doses of CP 55940)
preventing the increase of the resting time as observed
increased significantly standard chow intake (interaction
in control animals (Figure 4. C, D, and E, F). Finally, the
= 6.045 p<0.01; factor dose F
higher dose of WIN 55 212-2 did not alter the behavioral
p<0.001; factor hour F
= 54.66;p< 0.001) during the
profile observed in the control group. Nevertheless, animals
first hour. No statistically significant effect on the second
in this group spent more time eating during the beginning
period of observation was found (Figure 3).
of the first hour (Figure 4. G, H).
The behavioral profile produced by intra-NAcS injec-
tions of CP 55 940 (0.125, 0.25 and 0.5 nmol) was also assessed and it was found that all doses consistently de- layed the transition from eating to resting during the first hour of observation (after the 6th, 7th and 9th periods, respectively), with a corresponding increase on the time of activity (Figure 5. C, E, G). During the second hour of observation, the higher doses of CP 55 940 (0.25 and 0.5 nmol) decreased the time spent resting (Figure 5. F, H), while the lower dose (0.125 nmol) remained similar to the control group (Figure 5. D).
The present study was aimed to test the hypothesis that
acute activation of CB1R in the NAcS stimulate standard
Figure 3. Effects of intra-NAcS injections of different doses of CP
chow intake during the light phase of the photoperiod in
55 940 (CP) on standard chow intake during 2 behavioral records
rats maintained on ad libitum chow. It is shown here that
(hour 1 and hour 2). Data expressed in terms of means ± SEM.
both WIN 55 212-2 and CP 55 940 increase standard chow
***p<0.001 vs vehicle group; n= 4-6 per group.
intake and delay the expression of the natural satiety se- quence. According to the behavioral analyses carried out, intra-NAcS administration of CB1R agonists stimulates
food intake stimulated by cannabinoids
Figure 4. Behavioral satiety sequence of groups injected with vehicle (A, B), or WIN 55 212-2 0.125 (C, D), 0.25 (E, F) and 0.5 (G, H) nmol. Values are presented as mean (error bars represent SEM) of duration of each behavioral category (intake, resting, activity and groo- ming) during the two hours of the behavioral test (hour 1, hour 2). The vertical line (solid) indicates the moment in which the transition between intake and resting occurred; the vertical line (dotted) indicates that the transition between intake and resting occu rred before the beginning of the second behavioral record, n= 4-6 per group.
CORTÉS-SALAZAR, SUÁREZ ORTÍZ, CENDEJAS TREJO, MANCILLA-DÍAZ, LÓPEZ-ALONSO, ESCARTÍN-PÉREZ
Figure 5. Behavioral satiety sequence of groups injected with vehicle (A, B), or CP 55 940 0.125 (C, D), 0.25 (E, F) and 0.5 (G, H) nmol. Values are presented as mean (error bars represent SEM) of duration of each behavioral category (intake, resting, activity and grooming) during the two hours of the behavioral test (hour 1, hour 2). The vertical line (solid) indicates the moment in which the transition between intake and resting occurred; the vertical line (dotted) indicates that the transition between intake and resting occurred before the beginning of the second behavioral record, n= 4-6 per group.
food intake stimulated by cannabinoids
feeding during the light phase of the photoperiod, when
of food-induced behavioral alterations, such as food-
the basal intake is naturally low, suggesting that canna-
seeking. Supporting this notion, it was demonstrated that
binoid agonists consistently stimulate food consumption
the deletion of CB1R decreased operant behavior and the
when motivation for food is low and palatability is regular
motivation to obtain highly palatable food in CB1 KO
(standard chow).
mice (Guegan, et al., 2013). Furthermore, the findings that
Different studies have explored the role of cannabi-
CB1R KO rodents are resistant to diet-induced obesity
noid receptors in the hypothalamus (Verty, McGregor &
(Ravinet-Trillou, Delgorge, Menet, Arnone & Soubrié,
Mallet, 2005; Jamshidy & Taylor, 2001), indicating that
2004), and that over-activity of the endocannabinoid system
the stimulatory effect of cannabinoids is related to the
promotes an obese phenotype (Maccarrone, et al., 2010),
homeostatic modulation of food intake. Furthermore, be-
in conjunction with the cannabinoid-induced increase of
havioral experiments have shown that rimonabant bilaterally
activity observed in this study, suggest a functional con-
microinjected into the NAcS selectively decreased highly
nection between the endocannabinoid system, ineffective
palatable food intake in an operant task (Guegan, et al.,
inhibition of responses and obesity. Accordingly, chronic
2013). Our results confirm and extend the previous evidence
exposure to cannabinoid agonists should be studied in
that intra-accumbens administration of anandamide or 2-AG
order to determine if the cannabinoid-induced increase of
increase standard chow consumption (Soria-Gómez, et
activity is related to the impulsivity observed in obesity
al., 2007; Kirkham, et al., 2002) by a behavioral-specific
(Nederkoorn, Braet, Van Eijs, Tanghe & Jansen, 2006).
mechanism. Taking together these findings, it seems pos-
In spite of the pharmacological lack of selectivity of
sible that cannabinoid receptors modulate feeding behavior
WIN 55-212-2 and CP 55,940 at CB1 and CB2 receptors,
simultaneously by two independent processes; the first one,
there is no knowledge of reports of CB2 expression in the
homeostatic, characterized by affecting energy balance and
NAcS. Only multifocal expression of CB2 immunoreactiv-
motivational processes (hunger, appetite, satiety) (Cota, et
ity has been reported in different areas of rodent brains at
al., 2003; Escartín-Pérez, et al., 2009); and the second one,
levels much lower than those of CB1 receptors (cerebellum,
hedonic, increasing the rewarding properties of food and
hippocampus, olfactory tubercle, cerebral cortex, thalamus,
its hedonically positive sensory properties (Di Patrizio &
amygdala, basal ganglia, periaqueductal gray) (Gong, et al.,
Simansky, 2008).
2006). Nevertheless, conclusive pharmacological evidence
Despite the fact that the lower doses of WIN 55
of the CB1R participation in the reported effects should be
212-2 (0.125 and 0.5 nmol) and all doses of CP 55 940
provided (i.e. administering selective antagonists).
increased the time of activity (especially in the first hour
In summary, activation of CB1 receptors in the nucleus
of observation), this effect did not disrupt the behavioral
accumbens shell stimulates standard chow intake during the
profile associated to post-ingestive satiety sequence ei-
light phase of the photoperiod, when the basal food intake is
ther the transition from eating to resting, suggesting that
naturally low and the hedonically positive sensory properties
the endocannabinoid system in this particular conditions
of food may be stimulated. Further research should explore
promotes feeding and increases motor activity. It is well
in detail the relationship between the endocannabinoid
know that cannabinoids affect motor behavior via CB1R
system, impulsivity, and obesity.
by regulating glutamatergic and GABAergic transmission
(González, et al., 2009). Specifically, cannabinoid recep-
tors located in basal ganglia modulate both the inhibi-
tory and the excitatory neuronal transmission, providing
Bassareo, V. & Di Chiara, G. (1999). Modulation of feeding-
regulation of movement (Sanudo-Pena, Patrick, Patrick
induced activation of mesolimbic dopamine transmission
& Walker, 1996). Several reports showed that systemic
by appetitive stimuli and its relation to motivational state. European Journal of Neuroscience, 11(12), 4389-4397.
administration of the cannabinoid receptor agonist WIN 55,212-2 stimulated locomotor activity in the open field
Berner, L. A., Avena, N. M. & Hoebel, B. G. (2008). Bingeing,
self-restriction, and increased body weight in rats with li-
test at low doses (0.25-0.6 mg/kg) (Pandolfo, Pamplona,
mited access to a sweet-fat diet. Obesity (Silver Spring)
Prediger & Takahashi, 2007; Drews, Schneider & Koch,
2005) and this effect was described as a general motor
Cota, D., Marsicano, G., Tschöp, M., Grübler, Y., Flachskamm,
stimulating effect. Nevertheless, in the present study both
C., Schubert, M., Auer, D., Yassouridis, A., Thöne-Reine-
cannabinoid agonists were administered intra-NAcS and
ke, C., Ortmann, S., Tomassoni, F., Cervino, C., Nisoli, E.,
the behavioral profile elicited should not be considered
Linthorst, A. C., Pasquali, R., Lutz, B., Stalla, G. K. & Pa-
as a general stimulating effect, since the endogenous can-
gotto, U. (2003). The endogenous cannabinoid system affects
nabinoid system is strongly involved in the development
CORTÉS-SALAZAR, SUÁREZ ORTÍZ, CENDEJAS TREJO, MANCILLA-DÍAZ, LÓPEZ-ALONSO, ESCARTÍN-PÉREZ
energy balance via central orexigenic drive and peripheral li-
Melis, T., Succu, S., Sanna, F., Boi, A., Argiolas, A. & Melis,
pogenesis. Journal of Clinical Investigation, 112, 423-431.
M. R. (2007). The cannabinoid antagonist SR 141716A
Di Patrizio, N. V. & Simansky, K. J. (2008). Activating para-
(Rimonabant) reduces the increase of extra-cellular dopa-
brachial cannabinoid CB1 receptors selectively stimulates
mine release in the rat nucleus accumbens induced by a
feeding of palatable foods in rats. Journal of Neuroscience,
novel high palatable food. Neuroscience Letters, 419 (3),
28(39),9702-9709.
Dimitriou, S. G., Rice, H. B. & Corwin, R. L. (2000). Effects
Nederkoorn, C., Braet, B., Van Eijs, Y., Tanghe, A. & Jansen, A.
of limited access to a fat option on food intake and body
(2006). Why obese children cannot resist food: The role of
composition in female rats. International Journal of Eating
impulsivity. Eating Behaviors, 7, 315-322.
Disorders, 28,436-445.
Pandolfo, P., Pamplona, F. A., Prediger, R. D. & Takahashi, R.
Drews, E., Schneider, M. & Koch, M. (2005). Effects of the
N. (2007). Increased sensitivity of adolescent spontaneously
cannabinoid receptor agonist win 55,212-2 on operant be-
hypertensive rats, an animal model of attention deficit hype-
havior and locomotor activity in rats. Pharmacology Bio-
ractivity disorder, to the locomotor stimulation induced by
chemistry and Behavior, 80(1),145-150.
the cannabinoid receptor agonist WIN 55,212-2. European Journal of Pharmacology, 563(1–3), 141-148.
Escartín-Pérez, R. E., Cendejas-Trejo, N. M., Cruz-Martínez,
A. M., González-Hernández B., Mancilla-Díaz, J. M. &
Paxinos, G. & Watson, C. (1998). The brain in stereotaxic coor-
Florán-Garduño, B. (2009). Role of cannabinoid CB1 re-
dinates. New York: Academic Press.
ceptors on macronutrient selection and satiety in rats. Phy-
Perello, M., Chuang, J., Scott, M. M. & Lutter, M. (2010).
siology and Behavior, 96, 646-650.
Translational Neuroscience approaches to hyperphagia. The
Gardner, E. L. (2005). Endocannabinoid signaling system and
Journal of Neuroscience, 30(35), 11549-11554.
brain reward: Emphasis on dopamine. Pharmacology, Bio-
Quarta C., Bellocchio L., Manzini G., Mazza R., Cervino C.,
chemistry and Behavior, 81(2), 263-284.
Braulke L., Fekete C., Latorre R., Nanni C., Bucci M., Cle-
Gong, J. P., Onaivi, E. S., Ishiguro, H., Liu, Q. R., Tagliaferro,
mens L., Heldmaier G., Watanabe M., Leste-Lassere T.,
P. A., Brusco, A. & Uhla, G. R. (2006). Cannabinoid CB2
Maitre M., Tedesco L., FanelliF., Reuss S., KlausS., Sri-
receptors: Immunohistochemical localization in rat brain.
vastava R., Monory K., Valerio A., Grandis A., de Giorgio
Brain Research, 1071,10-23.
R., Pasquali R., Nisoli E., Cota D., Lutz B., Marsicano G. & Pagotto U. (2010). CB1 signaling in forebrain and sym-
González, B., Paz, F., Florán, L., Aceves, J., Erlij, D. & Flo-
pathetic neurons is a key Determinant of endocannabinoid
ran, B. (2009). Cannabinoid agonists stimulate [3H]-GABA
actions on energy balance. Cell Metabolism, 11, 273-285.
release in the globus pallidus of the rat when Gi protein- receptor coupling is restricted. Journal of Pharmacology
Ravinet-Trillou, C., Delgorge, C., Menet, C., Arnone, M. &
and Experimental Therapeutics, 328, 822-828.
Soubrié, P. (2004). CB1 cannabinoid receptor knockout in mice leads to leanness, resistance to diet-induced obesity
Guegan, T., Cutando, L., Ayuso, E., Santini, E., Fisone, G.,
and enhanced leptin sensitivity. International Journal of
Bosch, F., Martinez, A., Valjent, E., Maldonado, R. & Mar-
Obesity, 28, 640-648.
tina, M. (2013). Operant behavior to obtain palatable food modifies neuronal plasticity in the brain reward circuit. Eu-
Sanudo-Pena, M.C., Patrick, S. L., Patrick, R.L. & Walker, J.M.
ropean Neuropsychopharmacology, 23(2), 146-159.
(1996). Effects of intranigral cannabinoids on rotational be- havior in rats: Interactions with the dopaminergic system.
Jamshidy, N. & Taylor, D.A. (2001). Anandamide administra-
Neuroscience Letters, 206, 21-24.
tion into the ventromedial hypothalamus stimulates appetite in rats. British Journal of Pharmacology, 134, 1151-1154.
Soria-Gómez, E., Matías, I., Rueda-Orozco, P. E., Cisneros, M.,
Petrosino, S., Navarro, L. Di Marzo, V. & Próspero-García,
Kirkham, T. C., Williams, C. M., Fezza, F., & Di Marzo, V.
O. (2007). Pharmacological enhancement of the endocan-
(2002). Endocannabinoid levels in rat limbic forebrain and
nabinoid system in the nucleus accumbens shell stimulates
hypothalamus in relation to fasting, feeding and satiation:
food intake and increases c-Fos expression in the hypotha-
stimulation of eating by 2-arachidonoyl glycerol. British
lamus. British Journal of Pharmacology, 151, 1109-1116.
Journal of Pharmacology, 136(4), 550-557.
Verty, A.N., McGregor, I.S. & Mallet, P.E. (2005). Paraventri-
Maccarrone, M., Gasperi, V., Catani, M. V., Diep, T. A., Dai-
cular hypothalamic CB(1) cannabinoid receptors are invol-
nese, E., Hansen, H. S. & Avigliano, L. (2010). The endo-
ved in the feeding stimulatory effects of Delta(9)tetrahydro-
cannabinoid system and its relevance for nutrition. Annual
cannabinol. Neuropharmacology, 49 (8), 1101-1109.
Reviews of Nutrition, 30, 423-440.
Matias, I., Cristino, L. & Di Marzo, V. (2008). Endocannabinoi-
ds: Some like it fat (and sweet too). Journal of Neuroendo- crinology, 20(1), 100-109.
Source: http://editorial.ucatolica.edu.co/ojsucatolica/revistas_ucatolica/index.php/acta-colombiana-psicologia/article/download/165/205
UNDP Justice System Programme UNDP Justice System Programme Welcome to Timor- Leste Mai Ho Ksolok Iha Timor- Leste Partially compiled September 2003 UNDP Justice System Programme TABLE OF CONTENTS WELCOME TO TIMOR-LESTE.5 MAP OF TIMOR-LESTE.6 FACT SHEET ON TIMOR-LESTE.8 ADMINISTRATIVE DIVISION OF TIMOR-LESTE.9 BRIEF HISTORICAL OVERVIEW.10
HERZIENING RICHTLIJN HOGE BLOEDDRUK KWALITEITSINSTITUUT VOOR DE GEZONDHEIDSZORG CBO In samenwerking met: • Institute for Medical Technology Assessment• Nederlandsche Internisten Vereeniging• Nederlandse Hartstichting• Nederlandse Vereniging voor Cardiologie• Nederlands Huisartsen Genootschap• Nederlands Hypertensie Genootschap• Vereniging voor Epidemiologie




