Viagra gibt es mittlerweile nicht nur als Original, sondern auch in Form von Generika. Diese enthalten denselben Wirkstoff Sildenafil. Patienten suchen deshalb nach viagra generika schweiz, um ein günstigeres Präparat zu finden. Unterschiede bestehen oft nur in Verpackung und Preis.
Ijphs_july_aug_2008.indd
Herbal Excipients in Novel Drug Delivery Systems
A. SHIRWAIKAR*, ANNIE SHIRWAIKAR1, S. LAKSHMANA PRABU AND G. ARAVIND KUMAR
Department of Pharmaceutics, 1Department of Pharmacognosy, Manipal College of Pharmaceutical Sciences,
Manipal-576 104, India
Shirwaikar, et al.: Herbal excipients in NDDS
The use of natural excipients to deliver the bioactive agents has been hampered by the synthetic materials. However
advantages offered by these natural excipients are their being non-toxic, less expensive and freely available. The
performance of the excipients partly determines the quality of the medicines. The traditional concept of the
excipients as any component other than the active substance has undergone a substantial evolution from an inert
and cheap vehicle to an essential constituent of the formulation. Excipients are any component other than the active
substance(s) intentionally added to formulation of a dosage form. This article gives an overview of herbal excipients
which are used in conventional dosage forms as well as novel drug delivery systems.
Key words: Polysaccharides, volatile oils, controlled delivery
Excipients are primarily used as diluents, binders,
POLYSACCHARIDES IN
disintegrants, adhesives, glidants and sweeteners
in conventional dosage forms like tablets and capsules1. As the establishment of toxicity and
Natural polysaccharides are extensively used for the
approval from regulatory authorities poses a
development of solid dosage forms. These polymers
problem with synthetic excipients, of late more
of monosaccharides (sugars) are inexpensive and
interest is being shown by researchers in herbal
available in a variety of structures with a variety of
excipients. The drawback posed by heavy metal
properties. They are highly stable, safe, non-toxic,
contamination often associated with herbal and hydrophilic and gel forming in nature. Pectins, excipients is superseded by their lack of toxicity,
starch, guar gum, amylase and karaya gum are a few
easy availability, and economic considerations
polysaccharides commonly used in dosage forms.
in pharmaceutical industry as compared to their
Non-starch, linear polysaccharides remain intact in
synthetic counterparts. Present day consumers
the physiological environment of the stomach and
look for natural ingredients in food, drugs, and
the small intestine, but are degraded by the bacterial
cosmetics as they believe that anything natural
inhabitants of the human colon which make them
will be more safe and devoid of side effects.
potentially useful in targeted delivery systems to the colon2.
The traditional view that excipients are inert and do not exert any therapeutic or biological
action or modify the biological action of the drug
Pectins are non-starch, linear polysaccharides extracted
substance has changed and it is now recognized
from the plant cell walls. They are predominantly
that excipients can potentially influence the rate
linear polymers of mainly (1-4)-linked D-galacturonic
and/or extent of absorption of a drug. As herbal
acid residues interrupted by 1,2- linked L-rhamnose
excipients are non toxic and compatible, they have
residues with a few hundred to about one thousand
a major role to play in pharmaceutical formulation.
building blocks per molecule, corresponding to an
Hence, this paper is an attempt to review herbal
average molecular weight of about 50 000 to about
excipients used in NDDS.
1 80 0002. Being soluble in water, pectin is not able to shield its drug load effectively during its passage through the stomach and small intestine. It was found
that a coat of considerable thickness was required to
protect the drug core in simulated
in vivo conditions2.
July - August 2008
Indian Journal of Pharmaceutical Sciences
Hence the focus was shifted to the development of
has potential applications for the transdermal delivery
less soluble derivatives of pectin which get degraded
of chloroquine and perhaps in the management of
by the colonic microflora. Calcium salts of pectin
malaria. Calcium pectinate nanoparticles to deliver
reduced their solubility by forming an egg-box
insulin were prepared as a potential colonic delivery
configuration. To overcome the drawback of high
system by ionotropic gelation7. In relation to the food
solubility of pectin, mixed Þ lms of pectin with ethyl
industry, folic acid incorporated microcapsules were
cellulose were investigated as a coating material for
prepared using alginate and combinations of alginate
colon-specific drug delivery. These films combined
and pectin polymers so as to improve stability of
the colon specific degradation properties of pectin
folic acid. Folic acid stability was evaluated with
with the protective properties of the water insoluble
reference to encapsulation efficiency, gelling and
polymer ethyl cellulose2.
hardening of capsules, capsular retention during drying and storage. The blended alginate and pectin
Polymeric hydrogels are widely used as controlled-
polymer matrix increased the folic acid encapsulation
release matrix tablets. Sungthongjeen
et al.3
efÞ ciency and reduced leakage from the capsules as
investigated the high-methoxy pectin for its potential
compared to those made with alginate alone, they
value in controlled-release matrix formulations. The
showed higher folic acid retention after freeze drying
effects of compression force, ratio of drug to pectin,
and storage8.
and type of pectin on drug release from matrix tablets were also investigated. The results of the
in
In relation to cosmetics, using citronellal as a model
vitro release studies showed that the drug release
compound, pectin gel formulations were evaluated
from compressed matrix tablets prepared from pectin
for controlled fragrance release by kinetic and static
can be modified by changing the amount and the
methods. These formulations showed a prolonged
type of pectin in the matrix tablets. A very low
duration of fragrance release and limitation of
solubility pectin-derivative (pectinic acid, degree of
fragrance adsorption to the receptor skin layers.
methoxylation 4%) was found to be well suited as an
The increase in pectin concentrations suppressed
excipient for pelletisation by extrusion/spheronisation.
the fragrance release by a diffusion mechanism,
The capacity as an extrusion aid was found to be
thereby proving that pectin/calcium microparticles are
high; even formulations containing only 20% pectinic
promising materials for controlled fragrance release9.
acid resulted in nearly spherical pellets. All pectinic acid pellets were mechanically stable, had an aspect
ratio of approximately 1.15-1.20 and released 30-60%
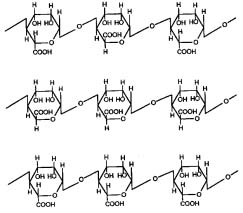
Alginates are natural polysaccharide polymers
of a low solubility model drug within 15 min both
isolated from the brown sea weed (Phaeophyceae).
in simulated gastric ß uid (0.1M HCl) and intestinal
Alginic acid can be converted into its salts, of
ß uid (phosphate buffer pH 6.8)4.
which sodium alginate is the major form currently used. A linear polymer consisting of D-mannuronic
Micro particulate polymeric delivery systems have
acid and L-guluronic acid residues arranged in
been suggested as a possible approach to improve the
blocks in the polymer chain, these homogeneous
low bioavailability characteristics shown by standard
blocks (composed of either acid residue alone) are
ophthalmic vehicles (collyria). In this context pectin
separated by blocks made of random or alternating
microspheres of piroxicam were prepared by the
units of mannuronic and guluronic acids. Alginates
spray drying technique.
In vivo tests in rabbits with
offer various applications in drug delivery, such as
dispersions of piroxicam-loaded microspheres also
in matrix type alginate gel beads, in liposomes, in
indicated a significant improvement of piroxicam
modulating gastrointestinal transit time, for local
bioavailability in the aqueous humour (2.5-fold) when
applications and to deliver the bio molecules in tissue
compared with commercial piroxicam eyedrops5.
engineering applications10 (Þ g. 1).
Musabayane
et al.6 investigated the suitability of
Bioadhesive sodium alginate microspheres of
amidated pectin as a matrix patch for transdermal
metoprolol tartrate for intranasal systemic delivery
chloroquine delivery in an effort to mask the bitter
were prepared to avoid the first-pass effect, as an
taste when orally administered. The results suggest
alternative therapy to injection, and to obtain improved
that the pectin-chloroquine patch matrix preparation
therapeutic efÞ cacy in the treatment of hypertension
Indian Journal of Pharmaceutical Sciences
July - August 2008
linking structure might control a highly water-soluble drug release for 24 h13.
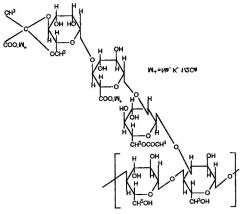
In a comparative study, alginate formulation appeared to be better than the polylactide-co-glycolide (PLG) formulation in improving the bioavailability of two clinically important antifungal drugs-clotrimazole and econazole. The nanoparticles were prepared by the emulsion-solvent-evaporation technique in case of PLG and by the cation-induced controlled geliÞ cation in case of alginate14.
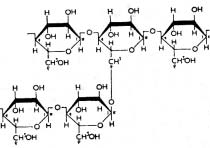
Starches:
It is the principal form of carbohydrate reserve in
green plants and especially present in seeds and
underground organs. Starch occurs in the form of
granules (starch grains), the shape and size of which
Fig. 1: Structure of alginic acid.
are characteristic of the species, as is also the ratio of
and angina pectoris. The microspheres were prepared
the content of the principal constituents, amylose and
using emulsification-cross linking method. In vivo
amylopectin. A number of starches are recognized for
studies indicated signiÞ cantly improved therapeutic
pharmaceutical use (Þ g. 2). These include maize (Zea
efficacy of metoprolol from microspheres, with
mays), rice (Oryza sativa), wheat (Triticum aestivum),
sustained and controlled inhibition of isoprenaline-
and potato (Solanum tuberosum)15.
induced tachycardia as compared with oral and nasal administration of drug solution11.
ModiÞ ed starch was tested for general applicability of a new pregelatinized starch product in directly
A new insert, basically consisting of alginates with
compressible controlled-release matrix systems. It
different hydroxyethylcellulose content was developed
was prepared by enzymatic degradation of potato
to maintain a constant drug level over a certain period
starch followed by precipitation (retrogradation),
in the eye, which cannot be achieved by conventional
Þ ltration and washing with ethanol. The advantages
eye drop application. This study showed good tolerance of the new calcium-alginate-insert applied to the ocular surface for controlled drug release12. In order to achieve 24 h release profile of water soluble drugs, sodium alginate formulation matrices containing xanthan gum or zinc acetate or both were investigated. The release of the drug from the sodium alginate formulation containing only xanthan gum was completed within 12 h in the simulated intestinal ß uid, while the drug release from the sodium alginate formulation containing only zinc acetate was completed within 2 h in the same medium. Only the sodium alginate formulation, containing both xanthan
gum and zinc acetate achieved a 24 h release proÞ le, either in the simulated intestinal ß uid or in the pH change medium (pH 1.2). The helical structure and high viscosity of xanthan gum possibly prevent zinc ions from diffusing out of the ranitidine HCL sodium alginate-xanthan gum-zinc acetate matrix so that zinc
ions react with sodium alginate to form zinc alginate
Fig. 2: Structures of (A) amylopectin or ∝- amylase and (B)
precipitate with a cross-linking structure. The cross-
July - August 2008
Indian Journal of Pharmaceutical Sciences
of the material include ease of tablet preparation,
the potential of a constant release rate (zero-order) for an extended period of time and its ability to
Gums are translucent and amorphous substances
incorporate high percentages of drugs with different
produced by the plants. Usually pathological products,
physicochemical properties. Release rates from
gums are produced when the plant is growing under
retrograded pregelatinized starch tablets can be
unfavorable conditions or when injured. Gums are
enhanced or decreased to the desired profile by
plant hydrocolloids and may be anionic or non ionic
different parameters like geometries of the tablet,
polysaccharides. On hydrolysis gums yield sugar and
compaction force and the incorporation of additional
salts of uronic acid20.
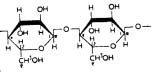
Guar gum:
To deliver proteins or peptidic drugs orally, Guar gum derived from the seeds of cyamopsis microcapsules containing a protein and a proteinase
tetragonolobus (Family Leguminosae) is a naturally
inhibitor were prepared. Starch/bovine serum albumin
occurring galactomannan polysaccharide. It is made
mixed-walled microcapsules were prepared using
up of a linear chain of β-D-mannopyranose joined
interfacial cross-linking with terephthaloyl chloride.
by β-(1-4) linkage with α-D-galactopyranosyl units
The microcapsules were loaded with native or amino-
attached by 1, 6- links in the ratio of 1:22 (Þ g. 3).
protected aprotinin by incorporating protease inhibitors in the aqueous phase during the cross-linking process.
Guar gum is used in colon-delivery systems due to
The protective effect of microcapsules with aprotinin
its drug release retarding property and susceptibility
for bovine serum albumin was revealed in vitro17.
to microbial degradation in the large intestine. Core tablets containing 5-aminosalisylic acid (5-ASA) were
Acetylating of starch considerably decreases its
prepared by wet granulation with starch paste and
swelling and enzymatic degradation. Thus, starch-
were compression coated with coating formulations
acetate (SA) based delivery systems were tested for
containing different quantities of guar gum The study
controlled drug delivery. It was proved that acetylation
conÞ rmed that selective delivery of 5-ASA to the colon
of potato starch can substantially retard drug release
can be achieved using guar gum as a carrier in the
by preparing and evaluating Þ lms of native starch and
form of a compression coating over the drug core21.
acetylated starch. Bovine serum albumin (BSA, mol. wt. 68 000), FITC-dextran (mol. wt. 4400), timolol
Further, guar gum-based matrix tablets of rofecoxib
(mol. wt. 332, log P=1.91) and sotalol-HCl (mol.
were prepared for their intended use in the
wt. 308, log P=-0.62) were used as model drugs.
chemoprevention of colorectal cancer. In vivo studies
All the model drugs were released rapidly from the
, prolonged absorption time
potato starch Þ lm in PBS pH 7.4 with and without
indicating that rofecoxib was
alpha-amylase in the dissolution medium (t50%
not released significantly in stomach and small
varied from 0.17 to 3.37 h). When compared to the
intestine, but was delivered to colon resulting in a
potato starch Þ lm, all the studied drugs were released
slow absorption of the drug and making it available
at a substantially slower rate from the SA films in
for local action in human colon22.
the corresponding media18. A comparative study was carried out to evaluate drug release from the SA microparticles (SA mps) and SA Þ lms.
The average degree of acetyl substitution (DS) per glucose residue in the starch was either 1.9 (SA DS 1.9) or 2.6 (SA DS 2.6). Timolol, calcein and BSA were used as model drugs. This study demonstrated the achievement of slow release of different molecular weight model drugs from the SA mps and films as compared to fast release from the native starch preparations19.
Fig. 3: Structure of guar gum.
Indian Journal of Pharmaceutical Sciences
July - August 2008
In an attempt to design oral controlled drug delivery
Gum arabic was used as an osmotic, suspending and
systems for highly water-soluble drugs using guar
expanding agent in the preparation of a monolithic
gum as a carrier in the form of three-layer matrix
osmotic tablet system (MOTS) with two oriÞ ces on
tablets, trimetazidine dihydrochloride was chosen as a
both side surfaces. Water-insoluble naproxen was
model drug because of its high water solubility. Both
selected as the model drug. The optimal MOTS
matrix tablets as well as three layer matrix tablets
was found to be able to deliver naproxen at a rate
were prepared and evaluated. The three-layer guar
of approximately zero order up to 12 h in pH 6.8.
gum matrix tablet provided the required release rate
Cumulative release at 12 h is 81%, and is independent
on par with the theoretical release rate for guar gum
of environment media and stirring rate. Therefore,
formulations meant for twice daily administration.
these MOTS can be used in the oral drug-controlled delivery Þ eld, especially for water-insoluble drugs28.
The results indicated that guar gum, in the form of three-layer matrix tablets, is a potential carrier in
Karaya gum:
the design of oral controlled drug delivery systems
Karaya gum is obtained from Sterculia urens (Family
for highly water-soluble drugs such as trimetazidine
sterculiaceae) is a partially acetylated polymer of
dihydrochloride23. The same study was carried out
galactose, rhamnose, and glucuronic acid26. Swellable
by using metoprolol tartrate a model drug with high
hydrophilic natural gums like xanthan gum and
solubility. The results indicated that guar gum, in the
karaya gum were used as release-controlling agents
form of three-layer matrix tablets, is a potential carrier
in producing directly compressed matrices. Caffeine
in the design of oral controlled drug delivery systems
and diclofenac sodium, which are having different
for highly water-soluble drugs such as metoprolol
solubilities in aqueous medium were selected as
model drugs. Gum erosion, hydration and drug release studies were carried out using a dissolution
Another water soluble drug, diltiazem HCl has given
apparatus (basket method) at two agitation speeds.
controlled release comparable with marketed sustained
In case of xanthan gum neither agitation speed nor
release diltiazem HCl tablets (D-SR tablets), which
drug solubility had any significant effect on water
are prepared in the form of matrix tablets with guar
uptake, but matrices with the lower proportion of gum
gum using the wet granulation technique25.
produced a lesser degree of hydration. In contrast, karaya gum displayed a much lower hydration
Gum acacia:
capacity and a higher rate of erosion, both markedly
Gum acacia or gum arabic is the dried gummy
affected by agitation speed. Hence it was concluded
exudate obtained from the stem and branches of
that drug release from xanthan and karaya gum
Acacia senegal (Linne) Willdenow and other related
matrices depended on agitation speed, solubility
species of acacia (Family Leguminosae). The gum
and proportion of drug. Both xanthan and karaya
has been recognized as an acidic polysaccharide
gums produced near zero order drug release with the
containing D-galactose, L-arabinose, L-rhamnose, and
erosion mechanism playing a dominant role, especially
D-glucuronic acid. Acacia is mainly used in oral and
in karaya gum matrices29. Park et al.30 showed that
topical pharmaceutical formulations as a suspending
mucoadhesive tablets prepared by karaya gum for
and emulsifying agent, often in combination with
buccal delivery, had superior adhesive properties as
tragacanth. It is also used in the preparation of
compared to guar gum and was able to provide
pastilles and lozenges and as a tablet binder26.
zero-order drug release, but concentrations greater than 50% w/w may be required to provide suitable
Sustained release of ferrous sulfate was achieved for 7
sustained release.
h by preparing gum arabic pellets. Release was further sustained for more than 12 h by coating the pellets
Xanthan gum:
with polyvinyl acetate and ethylene vinyl acetate,
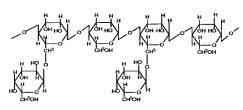
Xanthan gum is a high molecular weight extra cellular
respectively. An increase in the amount of gum arabic
polysaccharide produced by the fermentation of the
in the pellets decreased the rate of release due to the
gram-negative bacterium Xanthomonas campestris.
gelling property of gum arabic. The gel layer acts as
The primary structure of this naturally produced
a barrier and retards the rate of diffusion of FeSO
cellulose derivative contains a cellulosic backbone
through the pellet27.
(β-D-glucose residues) and a trisaccharide side chain
July - August 2008
Indian Journal of Pharmaceutical Sciences
of β-D-mannose-β-D-glucuronicacid-α-D-mannose
surface and the surface of fracture of tablets revealed
attached with alternate glucose residues of the main
that particles remained as coherent individual units
chain. The terminal D-mannose residue may carry
after compression process. Pellets showed close
a pyruvate function, the distribution of which is
compressibility degrees (49.9% for pellets comprising
dependent on the bacterial strain and the fermentation
diclofenac sodium and 48.5% for pellets comprising
conditions. The non-terminal D-mannose unit in the
ibuprofen). The release of the model drug from both
side chain contains an acetyl function. The anionic
type of tablets revealed different behaviours. Tablets
character of this polymer is due to the presence of
made of pellets comprising ibuprofen released the
both glucuronicacid and pyruvic acid groups in the
model drug in a bimodal fashion and the release
side chain26 (Þ g. 4).
behaviour was characterised as Case II transport mechanism (release exponent of 0.93). On the other
In one of the trials, xanthan gum showed a higher
hand, the release behaviour of diclofenac sodium
ability to retard the drug release than synthetic
from tablets made of pellets was anomalous (release
hydroxypropylmethylcellulose. Xanthan gum exponent of 0.70). For the latter case, drug diffusion and hydroxypropylmethylcellulose were used as
and erosion were competing mechanisms of drug
hydrophilic matrixing agents for preparing modiÞ ed
release tablets of diltiazem HCl. The amount of hydroxypropylmethylcellulose and xanthan gum
By utilizing retention properties of xanthan gum and
exhibited signiÞ cant effect on drug release from the
releasing properties of galactomannan, desire release
tablets prepared by direct compression technique.
profile was achieved in delivery of theophylline.
It was concluded that by using a suitable blend of
Hydrophilic galactomannan is obtained from the
hydroxypropylmethylcellulose and xanthan gum
seeds of the Brazilian tree Mimosa scabrella (Family
desired modiÞ ed drug release could be achieved31.
Leguminosae). The matrices made alone with xanthan gum (X) showed higher drug retention for
Compaction and compression properties of xanthan
all concentrations, compared with galactomannan (G)
gum pellets were evaluated and drug release from
matrices that released the drug too fast. The matrices
tablets made of pellets was characterized. Two types
prepared by combination of both gums were able to
of pellets were prepared by extrusion-spheronisation.
produce near zero-order drug release. The XG (conc
Formulations included xanthan gum, at 16% (w/w)
8%) tablets provided the required release rate (about
and diclofenac sodium or ibuprofen, at 10% (w/w)
90% at the end of 8 h), with zero-order release
among other excipients. Physical properties of pellets
and tablets were analysed. Laser proÞ lometry analysis and scanning electron microscopy of the upper
Tragacanth:
This gum is obtained from the branches of Astragalus
gummifer, Family Leguminosae20. Tragacanth when
used as the carrier in the formulation of 1- and 3-layer
matrices produced satisfactory release prolongation
either alone or in combination with other polymers34.
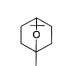
Volatile oils are generally mixtures of hydrocarbons and oxygenated compounds derived from these hydrocarbons. Many oils are terpenoid in origin; some of them are aromatic derivatives mixed with terpenes (e.g. cinnamon and clove). A few compounds (e.g. thymol and carvacrol) although aromatic in structure, are terpenoid in origin15 (Þ g. 5).
Fig. 4: Structure of xanthan gum.
Menthol is obtained by steam distillation of the
Indian Journal of Pharmaceutical Sciences
July - August 2008
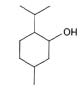
ß owering tops of Mentha piperita belonging to the
family Labiatae. A membrane-moderated transdermal
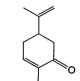
Caraway fruit consists of the dried, ripe fruits of
therapeutic system (TTS) of nimodipine using 2%w/w
Carum carvi (Umbelliferae). The volatile oil consists
hydroxypropylmethylcellulose (HPMC) gel as a
of the ketone carvone (fig. 5c) and the terpene
reservoir system containing menthol as penetration
limonene15 (Þ g. 5d). In another attempt, a limonene-
enhancer and 60%v/v ethanol-water as solvent
based transdermal therapeutic system (TTS) was
system was prepared. The in vivo evaluation of
prepared to study its ability to provide the desired
nimodipine TTS patch was carried out to find the
steady-state plasma concentration of nicorandil in
ability of the fabricated menthol-based TTS patch in
human volunteers. It was concluded that the limonene-
providing the predetermined plasma concentration of
based TTS of nicorandil provided the desired plasma
the drug in human volunteers. The results showed
concentration of the drug for the predetermined period
that the menthol-based TTS patch of nimodipine
of time with minimal fluctuations and improved
provided steady plasma concentration of the drug with
minimal ß uctuations with improved bioavailability in comparison with the immediate release tablet dosage
In a similar manner a carvone based and nerodilol
based transdermal therapeutic systems were prepared using nicorandil as a model drug. It was concluded
Menthol was tested for improving the bioavailability
that both TTS of nicorandil provided the desired in
of poorly water-soluble ibuprofen in the rectum with
vivo controlled-release profile of the drug for the
poloxamer. The effects of menthol and poloxamer
predetermined period of time39,40.
188 on the aqueous solubility of ibuprofen were investigated. The poloxamer gel with poloxamer
188 and menthol was found to be a more effective rectal dosage form for ibuprofen36. Terpenes such
Today the stress is on patient compliance and
as menthol (Þ g. 5a), cineole (Þ g. 5b) and propylene
to achieve this objective there is a spurt in the
glycol (PG) were tested as chemical enhancers to
development of NDDS. As the herbal excipients
improve the skin penetration of propranolol. Release
are promising biodegradable materials, these can
and skin permeation kinetics of propranolol from Þ lm
be chemically compatible with the excipients in
preparations were examined in in vitro studies using
drug delivery systems. In addition herbal excipients
a Franz-type diffusion cell. In vitro skin permeation
are non-toxic, freely available, and less expensive
studies showed that cineole was the most promising
compared to their synthetic counterparts. They have
enhancer among the enhancers examined37.
a major role to play in pharmaceutical industry. Therefore, in the years to come, there is going to be continued interest in the natural excipients to have better materials for drug delivery systems.
1. USP Subcommittee on excipients. Pharm Forum 1992;18:4387.
2. Sinha VR, Rachna K. Polysaccharides in colon speciÞ c drug delivery.
Int J Pharm 2001;224:19-38.
3. Sungthongjeen S, Pitaksuteepong T, Somsiri A, Sriamornsak P. Studies
on pectins as potential hydrogel matrices for controlled release drug delivery. Drug Develop Ind Pharm 1999;12:1271-6.
4. Tho I, Sande SA, Kleinebudde P. Pectinic acid: A novel excipient for
production of pellets by extrusion/spheronisation: Preliminary studies. Eur J Pharm Biopharm 2002;54:95-9.
5. Giunchedi P, Conte U, Chetoni P, Saettone MF. Pectin microspheres
as ophthalmic carriers for piroxicam: Evaluation in vitro and in vivo in albino rabbits. Eur J Pharm Sci 1999;9:1-7.
6. Musabayane CT, Munjeri O, Matavire TP. Transdermal delivery of
chloroquine by amidated pectin hydrogel matrix patch in the rat. Ren
Fig. 5: Structures of A. menthol, B. cineole, C. carvone and D.
7. Cheng K, Lim LY. Insulin-loaded calcium pectinate nanoparticles:
Effects of pectin molecular weight and formulation pH. Drug Develop
July - August 2008
Indian Journal of Pharmaceutical Sciences
Ind Pharm 2004;30:359-67.
8. Madziva H, Kailasapathy K, Phillips M. Alginate-pectin microcapsules
27. Batra V, Bhowmick A, Behera BK, Ray AR. Sustained release of
as a potential for folic acid delivery in foods. J Microencap
ferrous sulfate from polymer-coated gum arabica pellets. J Pharm Sci
9. Liu L, Chen G, Fishman ML, Hicks KB. Pectin gel vehicles for
28. Lu EX, Jiang ZQ, Zhang QZ, Jiang XG. A water-insoluble drug
controlled fragrance delivery. Drug Deliv 2005;12:149-57.
monolithic osmotic tablet system utilizing gum arabic as an osmotic,
10. Tonnesen HH, Karlssen J. Alginate in drug delivery systems Drug
suspending and expanding agent. J Control Release 2003;92:375-82.
Develop Ind Pharm 2002;28:621-30.
29. Munday DL, Cox PJ., Compressed xanthan and karaya gum matrices:
11. Rajinikanth PS, Sankar C, Mishra B. Sodium alginate microspheres of
Hydration, erosion and drug release mechanisms. Int J Pharm
metoprolol tartrate for intranasal systemic delivery: Development and
evaluation. Drug Deliv 2003;10:21-8.
30. Park CR, Munday DL. Evaluation of selected polysaccharide excipients
12. Fuchs-Koelwel B, Koelwel C, Gopferich A, Gabler B, Wiegrebe E,
in buccoadhesive tablets for sustained release of nicotine. Drug Develop
Lohmann CP. Tolerance of a new calcium-alginate-insert for controlled
Ind Pharm 2004;30:609-17.
medication therapy of the eye. Ophthalmologe 2004;101:496-9.
31. Gohel MC, Amin AF, Patel KV, Panchal MK. Studies in release
13. Zeng WM. Oral controlled release formulation for highly water-soluble
behavior of diltiazem HCl from matrix tablets containing
drugs: Drug--sodium alginate--xanthan gum--zinc acetate matrix. Drug
(hydroxypropyl) methyl cellulose and xanthan gum. Boll Chim Farm
Develop Ind Pharm 2004;30:491-5.
14. Pandey R, Ahmad Z, Sharma S, Khuller GK. Nano-encapsulation of
32. Santos H, Veiga F, Pina ME, Sousa JJ. Compaction compression and
azole antifungals: Potential applications to improve oral drug delivery.
drug release properties of diclofenac sodium and ibuprofen pellets
Int J Pharm 2005;301:268-76.
comprising xanthan gum as a sustained release agent. Int J Pharm
15. Trease GE, Evans WC editors. Text Book of Pharmacognosy, 15th ed.
London: Balliere, Tindall; 2002.
33. Vendruscolo CW, Andreazza IF, Ganter JL, Ferrero C, Bresolin TM.
16. Te-Wierik GH, Eissens AC, Bergsma J, Arends-Scholte AW, Bolhuis
Xanthan and galactomannan (from M. scabrella) matrix tablets for oral
GK. A new generation starch product as excipient in pharmaceutical
controlled delivery of theophylline. Int J Pharm 2005;296:1-11.
tablets, III: Parameters affecting controlled drug release from tablets
34. Siahi MR, Barzegar-Jalali M, Monaijemzadeh F, Ghaffari F, Azarmi
based on high surface area retrograded pregelatinized potato starch. Int
S. Design and evaluation of 1- and 3-layer matrices of verapamil
J Pharm 1997;157:181-7.
hydrochloride for sustaining its release. AAPSPharmSciTech
17. Larionova NV, Ponchel G, Duchene D, Larionova NI. Biodegradable
cross-linked starch/protein microcapsules containing proteinase inhibitor
35. Krishnaiah YS, Bhaskar P. Studies on the transdermal delivery of
for oral protein administration. Int J Pharm 1999;189:171-8.
nimodipine from a menthol-based TTS in human volunteers. Curr Drug
18. Tuovinen L, Peltonen S, Jarvinen K. Drug release from starch-acetate
Deliv 2004;1:93-102.
Þ lms. J Control Release 2003;91:345-54.
36. Yong CS, Yang CH, Rhee JD, Lee BJ, Kim DC, Kim DD. Enhanced
19. Tuovinen L, Peltonen S, Liikola M, Hotakainen M, Poso A, Jarvinen
rectal bioavailability of ibuprofen in rats by poloxamer 188 and
K. Drug release from starch-acetate microparticles and Þ lms with and
menthol. Int J Pharm 2004;269:169-76.
without incorporated alpha-amylase. Biomaterials 2004;25:4355-62.
37. Amnuaikit C, Ikeuchi I, Ogawara K, Higaki K, Kimura T. Skin
20. Kokate CK, Purohit AP, Gokhale SB, editors. Pharmacognosy, 22nd ed.
permeation of propranolol from polymeric film containing terpene
India: Nirali Prakashan; 2003. p. 133-66
enhancers for transdermal use. Int J Pharm 2005;289:167-78.
21. Krishnaiah YS, Satyanarayana S, Prasad YV. Studies of guar gum
38. Krishnaiah YS, Chandrasekhar DV, Rama B, Jayaram B, Satyanarayana
compression-coated 5-aminosalicylic acid tablets for colon-speciÞ c drug
V, Al-Saidan SM. In vivo evaluation of limonene-based transdermal
delivery. Drug Develop Ind Pharm 1999;25:651-7.
therapeutic system of nicorandil in healthy human volunteers. Skin
22. Al-Saidan SM, Krishnaiah YS, Satyanarayana V, Rao GS. In vitro and
Pharmacol Physiol 2005;18:263-72.
in vivo evaluation of guar gum-based matrix tablets of rofecoxib for
39. Krishnaiah YS, Al-Saidan SM, Chandrasekhar DV, Satyanarayana
colonic drug delivery. Curr Drug Deliv 2005;2:155-63.
V. Controlled in vivo release of nicorandil from a carvone-based
23. Krishnaiah YS, Karthikeyan RS, Gouri Sankar V, Satyanarayana V.
transdermal therapeutic system in human volunteers. Drug Deliv
Bioavailability studies on guar gum-based three-layer matrix tablets of
trimetazidine dihydrochloride in human volunteers. J Control Release
40. Krishnaiah YS, Al-Saidan SM, Chandrasekhar DV, Satyanarayana V.
Bioavailability of nerodilol-based transdermal therapeutic system of
24. Krishnaiah YS, Karthikeyan RS, Satyanarayana V. A three-layer
nicorandil in human volunteers. J Control Release 2005;106:111-22.
guar gum matrix tablet for oral controlled delivery of highly soluble metoprolol tartrate. Int J Pharm 2002;241:353-66.
25. Al-Saidan SM, Krishnaiah YS, Patro SS, Satyanaryana V. In vitro
and in vivo evaluation of guar gum matrix tablets for oral controlled
Accepted 5 July 2008
release of water-soluble diltiazem hydrochloride. AAPS PharmSciTech
Revised 01 March 2008
26. BhardwajTR, Kanwar M, Lal R, Gupta A. Natural gums and modiÞ ed
Received 22 January 2007
natural gums as sustained-release carriers. Drug Develop Ind Pharm
Indian J. Pharm. Sci., 2008, 70 (4): 415-422
Indian Journal of Pharmaceutical Sciences
July - August 2008
Source: http://www.annals.in/article.asp?issn=0250-474X;year=2008;volume=70;issue=4;spage=415;epage=422;aulast=Shirwaikar
a categorización de documentos es el proceso por el cual se asocian una o más categorías a textos escritos en un lenguaje natural basándose tan sólo en su L contenido. Aunque es posible construir de manera "manual" un categorizador, las técnicas estadísticas y, por tanto, automáticas son actualmente las preferidas puesto que no sólo ofrecen un rendimiento muy adecuado sino que resulta mucho más sencillo
Application Note PolarScreen Red™ (Invitrogen) Glucocorticoid Receptor Assay Tecan Infinite™ F500, Fluorescence Polarization The Glucocorticoid Receptor Assay description The Glucocorticoid Receptor (GR) belongs to the important Invitrogen has developed a variety of so called PolarScreenTM superfamily of ligand-activated, intracytoplasmatic











