Viagra gibt es mittlerweile nicht nur als Original, sondern auch in Form von Generika. Diese enthalten denselben Wirkstoff Sildenafil. Patienten suchen deshalb nach viagra generika schweiz, um ein günstigeres Präparat zu finden. Unterschiede bestehen oft nur in Verpackung und Preis.
Maxilofacial-sur.cl
Int J Clin Exp Med 2013;6(10):917-921
Original Article
Oral lichen planus treated with tacrolimus 0.1%
João Paulo Marinho Resende1, Maria das Graças Afonso Miranda Chaves1, Fernando Monteiro Aarestrup1,
Beatriz Vieira Aarestrup1, Sergio Olate2,3, Henrique Duque Netto1,4
1Master in Dental Science Program, Universidade Federal de Juis de Fora, Brazil; 2Center for Biomedical Re-
search, Universidad Autónoma de Chile, Chile; 3Division of Oral and Maxillofacial Surgery, Universidad de La
Frontera, Chile; 4Division of Oral and Maxillofacial Surgery, Universidade Federal de Juiz de Fora, BrazilReceived September 21, 2013; Accepted October 19, 2013; Epub October 25, 2013; Published October 30, 2013
Abstract: Oral lichen planus (OLP) is considered a chronic autoimmune inflammatory disease and its presence may
be related to increased emotional stress. The clinical relevance of OLP is the possibility of developing a squamous
cell carcinoma, the etiology of which is still unknown. The aim of this study is to treat OLP lesions resistant to con-
ventional treatment with corticosteroids, using topical tacrolimus 0.1% (Protopic®) twice a day for a period of eight
weeks. Fifteen patients were selected who had filled out a history form and a visual analog scale for pain before
and after treatment. All patients underwent an initial biopsy to diagnose the disease and another at the end of the
treatment period to evaluate the effect of the medication on the infiltrate. A weekly check was carried out, observing
the clinical appearance, pain symptoms and occurrence of side effects which, where present, were mild and tran-
sient. The results showed twelve patients (80%) with total or nearly total remission of pain symptoms and lesions,
two patients (13.33%) showed clearer lesions and only one patient (6.67%) had no change in clinical symptoms or
pain. Histopathological analysis showed OLP had a moderate or strong regression in twelve patients (80%) and an
absent or mild regression in three patients (20%). Based on these results, it was concluded that tacrolimus 0.1%
(Protopic®) is a safe and effective medication that improves the clinical appearance of the lesion, reduces pain as
well as the histopathological features of OLP.
Keywords: Oral lichen planus, tacrolimus, treatment, recalcitrant, corticosteroids
Topical tacrolimus is a powerful macrolide imm-
unosuppressant to prevent transplant rejetions
The prevalence of oral lichen planus (OLP) is
of organs such as the kidney, liver and heart [1,
relatively low (0.5% to 2.5%), being mainly asso-
3, 5, 9, 11]. It is a topical non-corticosteroidal
ciated with adult females at a ratio of 2:1, usu-
immunomodulator with a low adverse effect
al y emerging in the 4th to 5th decade of life, wh-
that presents a rapid response in the control of
ere 10% to 15% of the patients with OLP also
symptoms compared to traditional corticoste-
present cutaneous lesions [1-5].
roids [11-13].
The clinical presentation of OLP is interlaced
white lines on an erythroplastic base known as
The anti-inflammatory molecular mechanism of
"Wickham striae", these being symmetrical in
action of tacrolimus is similar to cyclosporine,
most patients [3, 4, 6]. It is thought that this
which inhibits the production of IL-2 by T lym-
reaction is mediated by T lymphocytes, where
phocytes [5, 14, 15] by inhibiting calcineurin
the cel s of the basal epithelial layer are recog-
phosphatase [6, 16], which in turn leads to the
nized as foreign due to the change in the anti-
inhibition of the nuclear gene transcription of
genicity of the surface of the cel s [2, 3, 7-9],
IL-2 cytosines and several other pro-inflamma-
which is why it is considered to be a disease of
tory cytosines such as IL4 and IL5 [17]. As a
unknown etiology and pathogeny occurring ma-
result, activation and differentiation of inflam-
inly in subjects with high stress levels [9, 10].
matory cel s such as T lymphocytes, eosino-
Currently OLP lesions are treated with different
phils or neutrophils are suppressed, which may
pharmacological options, and corticoids in their
explain why tacrolimus was also effective in
topical or systemic form are used frequently.
subjects with cicatricial pemphigoid.
Treatment of oral lichen planus
Table 1. Level of regression of the histopathological
The visual analog scale (VAS) was used in all
aspects between the first and second biopsy
the evaluations; this was completed by the
Regression of structural histological aspects (RHS) Level
patient, determining the degree of severity
Without regression
of the pain and the symptomatology. In the
eighth week a second biopsy was performed,
Moderate regression
fol owing the same protocol for col ection,
processing and diagnosis as the initial biop-
Strong regression
sy in order to assess the action of the drug in
relation to the histopathological aspects
The aim of this investigation is to analyze the
characteristic of OLP and also to confirm wheth-
behavior of tacrolimus 0.1% in subjects with
er the pain symptoms and clinical appearance
OLP who have undergone previous convention-
were related to the histopathological condi-
al treatments without favorable results.
tions. The scar from the first biopsy was used
as a reference to perform the new biopsy later-
Materials and methods
This research protocol was approved by the
A quantitative histopathological evaluation was
ethics committee of the Universidade Federal
made by two different observers to evaluate
de Juiz de Fora, Faculty of Dentistry. All the par-
the degree of regression or the condition of the
ticipants signed the informed consent and were
tissue. That analysis was made using a scale
informed of the scope of the study.
from 0 to 3, classifying each of the selected
15 patients (11 female and 4 male) aged betw-
segments (Table 1). Then the data were sub-
een 17 and 78 (age average 55 years) with sym-
jected to a descriptive statistical evaluation
ptoms associated with the disease were select-
through McNemar's test and a paired t-test or a
ed from the Department of Oral and Maxil ofacial
Wilcoxon test in case the variables did not
Surgery of the University Hospital of the UFJF
respond to conditions of normality considering
and the Neoplasia Diagnosis Support Service
a value of p<0.05 to achieve statistical rela-
of the UFJF. After the histopathological diagno-
sis of OLP, all the patients were treated with
20mg prednisolone for thirty days, and with
unfavorable results were incorporated into the
All the patients presented symptoms of the dis-
ease for at least 1 year of evolution prior to the
first therapeutic intervention. The most impor-
tant symptoms observed were a burning sensa-
One week prior to the start of the study, all
tion at the lesion site in 10 patients (3 patients
treatments were suspended for all the patients.
with burning and pain, 2 patients with itching,
An incisional biopsy had been performed previ-
and only one patient with burning, itching and
ously to confirm the histopathological diagnosis
pain at the same time). Three patients had only
of OLP. Each patient began the proposed treat-
pain and 2 only itching in the area.
ment with tacrolimus 0.1% (Protopic®) for two
months (8 weeks) using it in topical form as a
The visual analog scale before the treatment
cream twice a day (every 12 h). The subjects
with tacrolimus 0.1% (Protopic®) presented
were instructed to dry the place of application
variations from 4 to 9 (average 6.3) in the initial
and apply a fine layer using compressed cotton
phase. The most detailed analysis of the results
and not to eat for 1 h after the application.
showed that the severity of the symptoms in 7
patients was severe pain with levels of 8 (Table
During the treatment period, the patients were
evaluated weekly, recording the clinical appear-
ance of the lesion, the symptoms of the disease
After the first week of treatment unwanted
and the occurrence of side effects. The treat-
effects were described in 6 patients (40%), of
ment could be interrupted at any time that
which 2 patients had dry mouth, one patient
unwanted effects were determined by the
dry mouth and palatal changes and one patient
research group or when the patients indicated
reported burning at the application site; one
termination of the study.
patient presented dry mouth and a burning
Int J Clin Exp Med 2013;6(10):917-921
Treatment of oral lichen planus
Table 2. Responses of the visual analog scale (VAS) in the initial stage (before beginning the treat-
ment) and end stage (two months from beginning the treatment) of the 15 patients treated with
tacrolimus 0.1% Patients
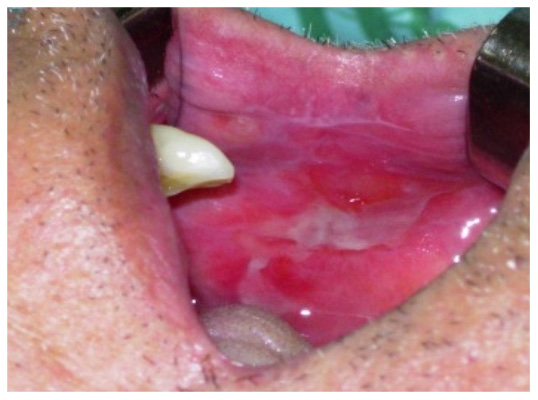
Figure 1. Lesion of erosive OLP in the mucosa; an
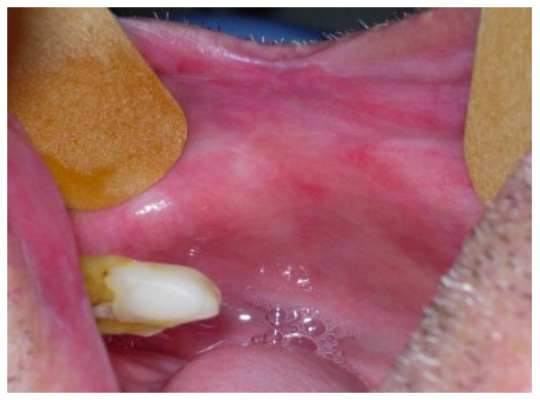
Figure 2. The same subject after 8 weeks of treat-
ulcerated leukoerythroblastic lesion with Wickham
ment demonstrating an oral mucosa free of lesions
striae in its periphery is observed.
and regression of the pathology.
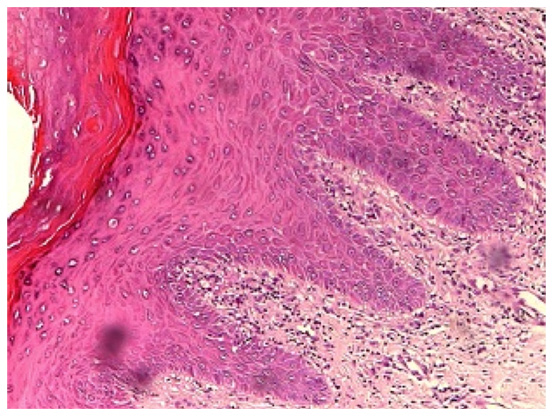
Figure 3. Histopathological analysis of the lesion
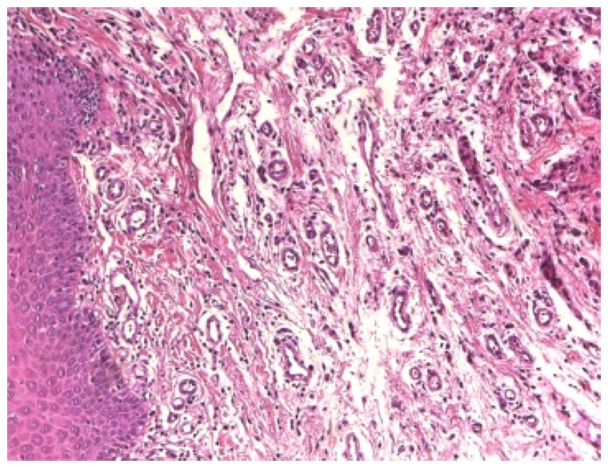
Figure 4. In the second biopsy (8 weeks later), a clean
where in the stage prior to treatment; epithelium is
oral mucosa was observed with regeneration of the
observed in serrated teeth, hyperkeratosis, hydropic
structural characteristics of the epithelium with the
degeneration of the base layer, acanthosis and pre-
almost total absence of inflammatory infiltrate (40X).
dominantly lymphocytic inflammatory infiltrate in
bundles of connective tissue (40X).
patients (40%) initial y reported a decrease in
OLP-associated symptoms, in five patients a
sensation and one patient reported only chang-
slight improvement was noted (33.3%) and in
es in the palatal area.
four patients (26.64%) no change was noted.
The clinical analysis showed no change.
It was not necessary to interrupt the treatment
for any of the subjects because the side effects
The maximum improvement was achieved in
were temporary and mild, disappearing com-
the 4th and 5th week of treatment, where 12
pletely during the study period; nine patients
subjects (79.92%) showed no type of pain or
reported no complications. In the first consulta-
indicated that the discomfort was less and
tion of the clinical study, after one week, six
often imperceptible (79.92%) (Figures 1 and 2).
Int J Clin Exp Med 2013;6(10):917-921
Treatment of oral lichen planus
In the clinical examination of the patients there
who had a recurrence of the disease with pain
were substantial improvements where the
symptoms received new pharmacological treat-
lesions disappeared completely or were almost
ment for the OLP.
imperceptible. Two patients were observed
who, despite indicating an improvement, still
presented clearer lesions; in one patient the
All the patients included in this study used daily
symptoms did not change and no changes in
doses of 20 mg prednisolone (once a day) for a
the clinical appearance were observed either.
period of at least 4 weeks before the initial
When the 8 weeks of the study were complete,
biopsy without obtaining satisfactory results or
a new VAS was conducted, presenting respons-
remission of signs or symptoms. This indication
es with a maximum variation between 0 and 4
was proposed by some authors [4, 18] where
with an average of 1.1 (Table 2), and when the
they reported that 20 mg prednisolone taken
initial comparison was set against the final one,
oral y may be effective in the treatment of OLP
it was observed that there was a significant dif-
without needing high doses to obtain a positive
ference (p<0.05) between the two scales
(p=0.001). Twelve patients presented no pain
There is a considerable number of patients who
symptoms or these were mild. In one patient
do not respond to conventional treatments,
who presented with moderate pain, this passed
which is why there is a need to find new thera-
to mild and in two patients the symptoms
peutic modalities to control OLP that have fewer
remained constant throughout the treatment.
side effects [1, 13, 18, 19].
Additional y, the statistical analysis of the data
showed that the improvement in symptoms
In this study, the topical application of tacroli-
was related to the clinical improvements in the
mus 0.1% was indicated due to advantages
OLP (p<0.05).
such as the reduction of side effects and fast
action in the control of symptoms [1, 12, 13].
Comparison of the biopsies taken towards the
The choice of the drug as a second line of treat-
end of the treatment with the initial biopsies
ment fol ows the direction taken by previously
revealed regression in the histopathological
published works [10, 11, 16-18, 20] that show
structures (RHS) of the OLP. This histopatho-
success in treatment with this drug.
logical situation, as with the subject's VAS, pre-
sented statistical y significant differences at
The dosage used was the manufacturer's rec-
the end of the treatment (p<0.05) (Figures 3
ommendation according to some studies [1, 5,
14] that showed this to be an efficient dosage;
however, others suggest that application of the
In terms of assessing the final clinical appear-
drug three times a day [21] or four times a day
ance of the lesions, the 12 patients (80.04%)
[15] may be more efficient. Additional y, the
with RHS of level 2 or 3 presented an almost
best results with tacrolimus 0.1% have been
total reduction or complete disappearance of
between the fourth and fifth week [1, 18, 19],
the lesion and with non-existent or mild symp-
which is also related positively to our results;
toms. In one patient (6.67%) with a RHS 1, the
others, however, show greater variation of this
clearest lesion was observed and had a sub-
time for treatment [10, 11]. Our results present-
stantial improvement in the pain symptoms
ed the most subjective and objective (clinical)
and of the two patients who had a RHS 0, one
improvements between the 4th and 5th week.
had the best defined lesion with the same
After 8 weeks there was an 80% improvement
symptoms as at the beginning of the treatment
where the lesion was practical y no longer per-
and the other had no changes in the clinical pic-
ceptible. Even so, two patients presented defi-
ture, pain or the histopathological structure.
ciencies in their evolution, similar to that obser-
After 5 months of evolution after the end of the
ved by other authors [21].
treatment, of the 12 patients who had lesions
Using a quantitative analysis, the close relation
that had practical y or completely disappeared,
between the improvement in clinical character-
two patients had a recurrence of the disease,
istics, improvement in symptoms and improve-
with one of these presenting a new, smal er yet
ment in histopathological characteristics was
asymptomatic lesion with total surgical resec-
observed [22], characterized by a reconstruc-
tion of the lesion, whereas the other patient
tion of the lining epithelium in the treated areas,
Int J Clin Exp Med 2013;6(10):917-921
Treatment of oral lichen planus
reducing epithelial hyperplasia and hyperkera-
[10] Lozada-Nur FI, Sroussi HY. Tacrolimus powder
tosis of the area.
in orabase 0.1% for the treatment of oral li-
chen planus and oral lichenoid lesions: An
Final y, in light of these results, the use of tacro-
open clinical trial. Oral Surg Oral Med Oral
limus 0.1% is efficient in the control of lesions
Pathol Oral Radiol Endod 2006; 102: 744-749.
and the symptoms associated with the OLP
[11] Donovan JC, Hayes RC, Burgess K, Leong IT,
that does not respond to other therapies with
Rosen CF. Refractory erosive oral lichen planus
corticosteroids. Studies with a greater number
associated with hepatitis C: Response to topi-
of subjects must be conducted to be able to
cal tacrolimus ointment. J Cutan Med Surg
2005; 38: 1-6.
recommend the use of this drug at different
[12] Byrd JA, Davis MD, Bruce AJ, Drage LA, Rogers
stages of OLP.
RS 3rd. Response of oral lichen planus to topi-
cal tacrolimus in 37 patients. Arch Dermatol
Disclosure of conflict of interest
2004; 140: 1508-1512.
[13] Bruce A, Rogers RS 3rd. New and old thera-
peutics for oral ulcerations. Arch Dermatol
2007; 143: 519-523.
Address correspondence to: Dr. Sergio Olate,
[14] Olivier V, Lacour JP, Mousnier A, Garraffo R,
Facultad de Odontología, Universidad de La
Monteil RA, Ortonne JP. Treatment of chronic
Frontera, Claro Solar 115, 4to Piso, Oficina 20,
erosive oral lichen planus with low concentra-
Temuco, Chile. E-mail:
tions of topical tacrolimus: An open prospec-
tive study. Arch Dermatol 2002; 138: 1335-
[15] Rozycki TW, Rogers RS 3rd, Pittelkow MR, McE-
[1] Vente C, Reich K, Rupprecht R, Neumann C.
voy MT, el-Azhary RA, Bruce AJ, Fiore JP, Davis
Erosive mucosal lichen planus: response to
MD. Topical tacrolimus in the treatment of
topical treatment with tacrolimus. Br J Derma-
symptomatic oral lichen planus: A series of 13
tol 1999; 140: 338-342.
patients. J Am Acad Dermatol 2002; 46: 27-
[2] Ling MR. Topical tacrolimus and pimecrolimus:
future directions. Semin Cutan Med Surg
[16] Nasr IS. Topical tacrolimus in dermatology. Clin
2001; 20: 268-277.
Exp Dermatol 2000; 25: 250-254.
[3] Edwards PC, Kelsch R. Oral lichen planus: clin-
[17] Fuchs M, Schliemann-Willers S, Heinemann C,
ical presentation and management. J Can
Elsner P. Tacrolimus enhances irritation in a
Dent Assoc 2002; 68: 494-499.
5-day human irritancy in vivo model. Contact
[4] Scully C, Eisen D, Carrozzo M. Management of
Dermatitis 2002; 46: 290-294.
oral lichen planus. Am J Clin Dermatol 2000;
[18] Assmann T, Becker J, Ruzicka T, Megahed M.
Topical tacrolimus for oral cicatricial pemphi-
[5] Gupta AK, Adamiak A, Chow M. Tacrolimus: a
goid. Clin Exp Dermatol 2004; 29: 674-676.
review of its use for the management of der-
[19] Shichinohe R, Shibaki A, Nishie W, Tateishi Y,
matoses. J Eur Acad Dermatol Venereol 2002;
Shimizu H. Successful treatment of severe re-
16: 100-114.
calcitrant erosive oral lichen planus with topi-
[6] Becker JC, Houben R, Vetter CS, Bröcker EB.
cal tacrolimus. J Eur Acad Dermatol Venereol
The carcinogenic potential of tacrolimus oint-
2006; 20: 66-8.
ment beyond immune suppression: a hypoth-
[20] Fleischer AB Jr. Treatment of atopic dermatitis:
esis creating case report. BMC Cancer 2006
Role of tacrolimus ointment as a topical non-
Jan 11; 6: 7.
corticosteroidal therapy. J Allergy Clin Immunol
[7] Torti DC, Jorizzo JL, McCarty MA. Oral lichen
1999; 104: S126-30.
planus: A case series with emphasis on thera-
[21] Johnson H, Soldano AC, Kovich O, Long W. Oral
py. Arch Dermatol 2007; 143: 511-515.
lichen planus. Dermatol Online J 2008 May
[8] Sugerman PB, Savage NW, Walsh LJ, Zhao ZZ,
Zhou XJ, Khan A, Seymour GJ, Bigby M. The
[22] Maeda H, Reibel J, Holmstrup P. Keratin stain-
pathogenesis of oral lichen planus. Crit Rev
ing pattern in clinically normal and diseased
Oral Biol Med 2002; 13: 350-365.
oral mucosa of lichen planus patients. Scand J
[9] Loeb I, Daelemans A, André J. Lichen plan buc-
Dent Res 1994; 102: 210-215.
cal érosive et onguent de tacrolimus. Rev Sto-
matol Chir Maxillofac 2005; 106: 1-5.
Int J Clin Exp Med 2013;6(10):917-921
Source: http://maxilofacial-sur.cl/uploads/files/T18.2013.pdf
POLICY & PROCEDURE TABLE OF CONTENTS UTILIZATION MANAGEMENT Day Surgery Pre-Authorization Delivery & Length of Stay Emergency Admissions Inpatient Transfer Home Visit For 1 Day OB Stay Elective & Non-Emergency Admissions Prior Day Admissions Psychiatric & AODA Admissions CT, MRI Prior Authorizations PT, OT, & ST Prior Authorizations
Spectro Analytic Irradia AB Exempel på rct studier under 2007 Från www.pubmed.com / sökord laser therapy 1: Tanzan Health Res Bull. 2007 Sep;9(3):196-201. Links Low intensity laser therapy is comparable to bromocriptine-evening primrose oil for the treatment of cyclical mastalgia in Egyptian females. Saied GM, Kamel RM, Dessouki N.







